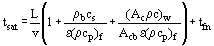Fixed beds are used in the process industries as catalytic chemical reactors, adsorption/desorption beds, thermal regenerators, heat storage devices and pebble bed heaters. In all of these operations, the transfer of heat and mass takes place between the fluid and solid phases and the transfer can be either steady state or transient. The solid phase can be in several forms: randomly dumped packings, ordered mono-sized particles and monolithic blocks of various geometrical shapes. The fixed bed geometry itself is normally cylindrical and the flow of the fluid through the bed is parallel to the axis of the cylinder. Radial flow through annular fixed beds is also used when low pressure drop restrictions are specified.
The major design parameters are the pressure drop across the fixed bed, and the heat and mass transfer coefficients between the fluid and the surface of the solid phase. Diffusion of heat and mass into the interior of the solid phase can be a significant mechanism of transfer, but it is common to employ lumped transfer coefficients at the surface to account for the internal diffusion, and to use average solid temperatures and concentrations in the design calculations. Pressure drop across a fixed bed is calculated from empirical formulas: the most common formula being that proposed by Ergun in 1952, i.e.,
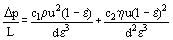
where Δp is the pressure drop, L is the length of the fixed bed, d is the equivalent diameter of the particle, defined as the equivalent volume sphere (= 6 × volume/surface area), ε is the bed voidage (porosity = free volume/total volume), u is the superficial velocity based on flow through an empty fixed bed, η is the fluid viscosity and c1 and c2 are correlation values obtained by regression of experimental data. Universal values of c1 and c2 do not exist, see Heggs (1983), although, for randomly packed spherical particles, the values obtained by MacDonald et al. (1979) (c1 = 1.8 and c2 = 180) provide reasonable predictions of the pressure drop over a wide Reynolds number range: 0.1 to 10000, Otherwise, it is essential that they are evaluated from experimental data of the fixed bed under consideration. The measurement of the bed voidage e is crucial, because Eq. (1) is very sensitive to this parameter. The value of the equivalent volume sphere, d, is straightforward for mono-sized particles, but in many process systems a mixture of particle sizes, and occasionally shapes, is used for the fixed bed. A suitable weighted mean of the equivalent volume sphere must be used in Eq. (1), [Hawkins (1993)].
For the calculation of the pressure drop across annular fixed beds, it is necessary to take into account the direction of the radial flow and the effects of the fluid momentum changes, i.e.,
where
 , where
, where
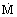 is the total mass flow rate and L the height of the annulus, Di and D0 are the inner and outer diameters of the annular fixed bed. The positive sign is for flow inwards and the negative sign is for flow outwards. Equation (2) assumes that the flow is evenly distributed along the length and that the inlet and outlet manifolds do not effect the total pressure drop of the annular system. However, flow maldistribution can occur in annular systems due to the pressure profiles in the inlet and outlet manifolds [Heggs et al. (1994b)]; this will result in larger overall pressure drops than the value predicted by Eq. (2). Two further relevant physical properties of the fixed beds are required for transfer processes: surface area, Av, per unit bed volume
is the total mass flow rate and L the height of the annulus, Di and D0 are the inner and outer diameters of the annular fixed bed. The positive sign is for flow inwards and the negative sign is for flow outwards. Equation (2) assumes that the flow is evenly distributed along the length and that the inlet and outlet manifolds do not effect the total pressure drop of the annular system. However, flow maldistribution can occur in annular systems due to the pressure profiles in the inlet and outlet manifolds [Heggs et al. (1994b)]; this will result in larger overall pressure drops than the value predicted by Eq. (2). Two further relevant physical properties of the fixed beds are required for transfer processes: surface area, Av, per unit bed volume

and the bulk density—mass per volume of the system,

Heat transfer in fixed beds can be either a steady state or a transient process and various mathematical models have been proposed to describe transfer of heat between the fliud flowing through the fixed bed and the packing of the fixed bed. In some process applications there is transfer from the fluid to and across the wall of the container of the fixed bed. The definitive paper of Amundson (1956) contains almost all of the models needed to describe the transfer of heat in fixed beds. Without exception, it is necessary to solve coupled differential equations, either ordinary or partial. If chemical reactions or sorption processes are taking place in the fixed bed, then the system of equations becomes nonlinear. Space precludes a detailed discussion of all the possible models and solutions. Only the transient nonreacting and nonsorptive systems will be discussed here. This corresponds to several applications: heat storage devices, pebble bed heaters, cooling of desorption beds, start-up and shut-down of process equipment and pipelines. The level of sophistication of the calculation is directly related to the number of parameters required for the evaluation of the solution pertaining to the problem.
The simplest representation of the transient heating or cooling of a fixed bed corresponds to the assumption that the film heat transfer coefficient between the fluid and the solids is infinite and the temperatures throughout the fixed bed are identical [Heggs (1994a)]. If the fixed bed is initially at a uniform temperature. T0, and the inlet fluid temperature to the bed is changed to Tfi by some forcing function, fn(t), then the time for the fixed bed and its container to equilibrate at the inlet fluid temperature is given by
where v is the interstitial velocity, = u/ε, and Acb and Acw are the cross-sectional areas of the empty fixed bed and container wall, respectively, and tfn is the time for the forcing function of the inlet fluid temperature to reach the value Tfi. If the forcing function is a step change, then tfn is zero. The first term in the brackets of Equation (5) is the residence time of the fluid in the fixed bed, the second and third terms represent the times for the packing and the container wall to equilibrate. Normally it is assumed that the fixed bed is adiabatic with respect to the container wall, so that the third term in Equation (5) is neglected. This is often the cause of the underprediction of the time for equilibrium.
The time to saturation is also delayed when the fluid to solids heat transfer coefficients are finite. In this representation of the transfer processes, the temperatures of the phases are different during the transient period of operation and it is impossible to present an explicit formula to predict the time of heating or cooling. The equations describing the system are as follows: Heat balance on the fixed bed

this balance ignores any thermicity effects of chemical reaction or the sorption process—no heat of adsorption or desorption. This equation becomes the pseudo-homogeneous system, if α → ∞ and the container wall effects are ignored. For finite values of the heat transfer coefficient, a rate equation is required to couple the temperatures between the fluid and solid phases, as follows:

Initial condition:
Boundary condition:

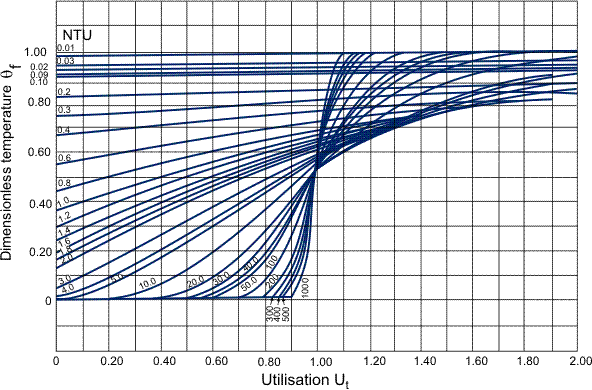
The outlet fluid temperature, Tf0 of the above system of Equations (5) to (8) is a function of two dimensionless groups: the Number of Transfer Units

and the utilization factor

The following graph, Figure 1, shows the response of the dimensionless outlet fluid temperature, θf (={Tf0 - T0}/{Tfi - T0}), over the range of NTU values: 0.01 to ∞ for Ut from 0 to 2.0. The forcing function for these results is a step change in the fluid inlet temperature.
The delay caused by the finite heat transfer between the fluid and the solid depends upon the value of NTU and, if this goes to infinity, then the vertical line at Ut = 1.0 corresponds to Equation (5) without the wall term. The literature abounds with correlations for the prediction of the fluid to solid heat transfer coefficient, but which should be used to predict the value of the coefficient remains a bone of contention. Foumeny and Ma (1991) list a number of correlations for randomly dumped fixed beds of spherical particles and recommend the use the equation proposed by Abou-Ziyan (1988):
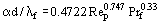
where the Reynolds number is defined as

The response of annular fixed beds can be predicted by the same method as above if the dimensionless groups NTU and Ut are redefined as follows:



The effects of other mechanisms of transfer in the fixed beds can be accommodated by using a lumped heat transfer coefficient, αzℓ:

where the second term accounts for conduction in a spherical particle and the third term accounts for axial dispersion as the fluid flows through the fixed bed. The Peclet Number, Ped, can be predicted from the following empirical equation:
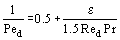
where Red is the Reynolds Number based on particle diameter and Pr is the Prandtl Number.
REFERENCES
Abou-Ziyan, Z. Z. H. (1988) Heat and Momentum Transfer in Porous Material Used for Thermal Energy Storage, PhD Thesis, University of Leeds, UK.
Amundson, N. R. (1956) Solid-Fluid Interactions in Fixed and Moving Beds, Ind. Eng. Chem., 49(1)26-43.
Foumeny, E. A. and Ma, J. (1991) Design Correlations for Heat Transfer System, Chapter 11, Heat Exchange Engineering: Volume 1, Design of Heat Exchangers, Ellis Horwood Limited, London, 159-178.
Hawkins, A. E. (1993) The Shape of Powder-Particle Outlines, RSP Series—Materials Science and Technology Series No. 1, John Wiley and Sons, London.
Heggs, P. J. (1994a) Heat Transfer in Particulate Systems—The Infamous Film Heat Transfer Coefficient. Heat Transfer 1994—Proceedings of the Tenth International Heat Transfer Conference, Vol. 1, IChemE, Rugby, UK, 461-466.
Heggs, P. J., Ellis, D. I. and Ismail, M. S. (I994b) The Modeling of Fluid Flow Distributions in Annular Packed Beds, Gas Separation and Purification, 8, 4, 257-264. DOI: 10.1016/0950-4214(94)80006-5
Heggs, P. J. (1983) Fixed Beds, Section 2.2.5, Heat Exchanger Design Handbook, 2, Hemisphere Publishing Corporation, N.Y., 2.2.5-1-5.
MacDonald, I. F., El-Sayed, M. S., Mow, K., and Dullen, F. A. L. (1979) Flow Through Porous Media—the Ergun Equation Revised, Ind. Eng. Chem. Fund, 18, 198.
References
- Abou-Ziyan, Z. Z. H. (1988) Heat and Momentum Transfer in Porous Material Used for Thermal Energy Storage, PhD Thesis, University of Leeds, UK.
- Amundson, N. R. (1956) Solid-Fluid Interactions in Fixed and Moving Beds, Ind. Eng. Chem., 49(1)26-43. DOI: 10.1021/ie50553a016
- Foumeny, E. A. and Ma, J. (1991) Design Correlations for Heat Transfer System, Chapter 11, Heat Exchange Engineering: Volume 1, Design of Heat Exchangers, Ellis Horwood Limited, London, 159-178.
- Hawkins, A. E. (1993) The Shape of Powder-Particle Outlines, RSP Series—Materials Science and Technology Series No. 1, John Wiley and Sons, London.
- Heggs, P. J. (1994a) Heat Transfer in Particulate Systems—The Infamous Film Heat Transfer Coefficient. Heat Transfer 1994—Proceedings of the Tenth International Heat Transfer Conference, Vol. 1, IChemE, Rugby, UK, 461-466.
- Heggs, P. J., Ellis, D. I. and Ismail, M. S. (I994b) The Modeling of Fluid Flow Distributions in Annular Packed Beds, Gas Separation and Purification, 8, 4, 257-264. DOI: 10.1016/0950-4214(94)80006-5
- Heggs, P. J. (1983) Fixed Beds, Section 2.2.5, Heat Exchanger Design Handbook, 2, Hemisphere Publishing Corporation, N.Y., 2.2.5-1-5.
- MacDonald, I. F., El-Sayed, M. S., Mow, K., and Dullen, F. A. L. (1979) Flow Through Porous Media—the Ergun Equation Revised, Ind. Eng. Chem. Fund, 18, 198.