Chromatography is a physicochemical differential-migration method of separation, analysis and investigation of substances. Credit for discovering the chromatographic method belongs to Russian botanist M.S. Tsvet (1903). According to him, the prerequisite for separating components of a mixture is a different kind of adsorption. Modern chromatography employs separation of substances, apart from molecular adsorption, and other physicochemical phenomena.
The term "chromatography" refers to all kinds of separation based on the division of components of the analyzed mixture between an eluent phase that can be a gas (vapor) or a liquid, and an eluate phase that can be a solid or a liquid. Different equilibrium and kinetic distribution of substances between them are prerequisites of chromatographic separation.
Depending on the phases of the substances involved, a distinction is made between gas and liquid chromatography. Gas chromatography includes gas-adsorption (gas-solid) and gas-liquid chromatography. Liquid chromatography is subdivided into liquid-liquid, liquid-solid and liquid-gel chromatography. The former characterizes the phase state of the eluent; the second, that of the eluate.
Chromatography is classified by the separation mechanism, that is the nature of interaction between a sorbent (an absorbing substance) and a sorbate (a substance being absorbed) as follows:
Adsorption chromatography, in which separation is based on the different adsorptive properties of mixture components separated by a solid adsorbent;
Partition chromatography, separation based on the different solubility of substances separated in eluates (gas chromatography) as well as in liquid eluents and eluates;
Ion exchange chromatography, in which the different capabilities of mixture components in ion exchange are used as basis for separation;
Penetration chromatography, in which separation occurs due to the different molecular size and shape of separated substances, e.g., in molecular sieves.
Other types include precipitation chromatography (separation based on precipitation of substances with different solubility with a sorbent), adsorption and chelation chromatography (separation based on formation of coordination compounds of different strength on an adsorbent surface). Thus, as a rule, separation of substances proceeds by several mechanisms.
Depending on the geometry of an eluate's sorption layer and correspondingly, the equipment used, chromatography is subdivided into column and one-dimensional chromatography. The latter includes paper chromatography, in which separation of substances takes place on a special paper and thin-layer chromatography, in which separation is by virtue of a thin layer of sorbent. Column chromatography is used to separate a mixture in special columns, as in capillary chromatography. Column and thin-layer chromatography employ any separation mechanism, while paper chromatography uses partition and ion exchange mechanisms.
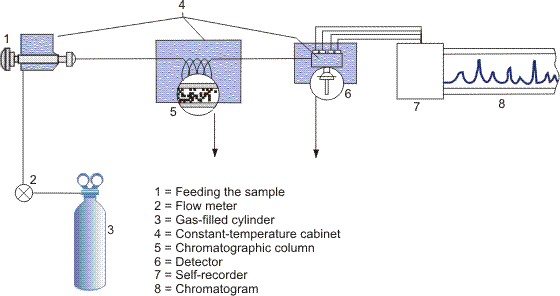
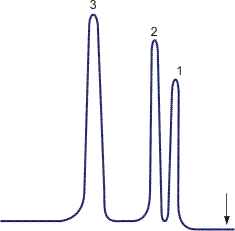
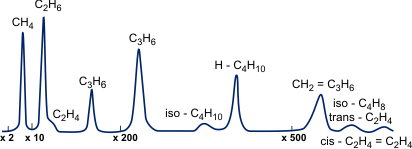
Chromatography is performed using special devices known as chromatographs which have, as main components, a chromatographic column and a detector (Figure 1). Thermal conductivity, electron capture, spectral (flame-photometric), density and other types of detectors are usually used. When the sample is fed, the mixture of substances is at the inlet to the chromatograph column. Eluent flow makes the mixture components migrate at different velocities due to the different adsorbing capabilities of the adsorbent surface. A detector at the column outlet continuously determines the concentration of separated compounds in the eluent. The detector signal, as a rule, is self-recorded and a chromatogram is obtained (Figures 2 and 3).
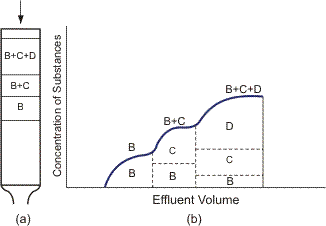
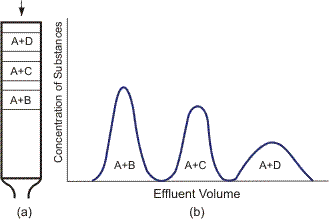
The three techniques for obtaining chromatograms are: frontal, elution and displacement. When the frontal technique is used, the eluent containing several components is continuously fed to the column. The substance derived at the column outlet is known as an effluent. For instance, if the eluent contains three substances B, C and D with the sorption ability order B < C < D, then separation in the column and on the chromatogram can be represented schematically (Figure 4).
Elution chromatography is the most commonly encountered. The chromatograph column is first washed by the eluent flow, the eluent having lower sorption power than any of the separated substances. Then the analyzed mixture is added at regular intervals into the eluent flow, the mixture in the column being separated into components with eluent zones between them. For instance, a mixture contains substances B, C and D with the sorption ability order B < C < D and the substance A is an eluent, its sorption ability being lower than that of B, i.e., A < B < C < D. Separation of substances in the column and on the chromatogram is shown in Figure 5.
When displacement chromatography is used, a small amount of mixture is fed to the column after which a displacer that is adsorbed better than any other mixture components is continuously passed through it. This results in the appearance of adjoining zones of separated substances.
Figure 6 schematically shows displacement chromatography where B, C and D are the separated substances; E is the displacer; and B < C < D < E, the sorption ability order. The frontal and displacement techniques require regeneration of the column before the next experiment is started.
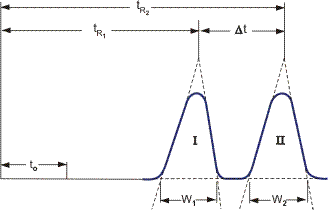
The main parameters governing chromatography are retention, efficiency and separation degree. They can be determined from a chromatogram. One of the basic characteristics of substance separation is the retention time of a given component tR (Figure 7), that is, the time elapsed from the moment the sample is fed to the column up to the moment the chromatographic zone of the substance output reaches its maximum.
An important factor in chromatography for a given substance is retained volume VR, which is determined by the equation
 , where
, where
 is the volume velocity of the flow. Peak sharpness depends on column efficiency, and the distance between the maxima is determined by its selectivity. Column efficiency is understood as a restricted washout of the chromatographic zone in the column. Efficiency can be expressed quantitatively by the number of "thermal trays" N into winch the chromatographic column is divided
is the volume velocity of the flow. Peak sharpness depends on column efficiency, and the distance between the maxima is determined by its selectivity. Column efficiency is understood as a restricted washout of the chromatographic zone in the column. Efficiency can be expressed quantitatively by the number of "thermal trays" N into winch the chromatographic column is divided

where σ is the standard deflection of the peak (σ = L/4). The length H of column in which an equilibrium between the substance concentrations in the eluent and the eluate is established is called the height of the equivalent theoretical tray H = L/N, where L is the column length. The overall height H of the tray is made up of components due to the nonhomogeneous flow of the eluent Hp, molecular diffusion Hd, mass exchange in the eluent Hm and in the eluate Hs Thus, H = Hp + Hd + Hm + Hs.
Selectivity is a common measure of substance separation during chromatography and a measure of relative retention. Selectivity is determined by:

where D2 and D1 are the separation coefficients of the second and the first mixture components, respectively. D = Cs/Cm, where Cs and Cm are the substance concentrations in eluates and eluents, respectively. Thus, the separation of mixture components is a function of D, Vs, H, and L.
A high rate and a high degree of separation make it possible to apply chromatographic techniques for qualitative and quantitative analyses of organic and inorganic compounds in scientific research and industrial processes. The main disadvantage of chromatographic techniques is the cyclic character of the analysis.
Chromatography is most frequently used for determining the concentrations and physicochemical properties of substances and for deriving high-purity chemical elements and complex compounds. Chromatographis techniques are widely used in controlling and automating production processes in the fields of environmental monitoring, agriculture, medicine, geology, etc.
REFERENCES
Perry, S., Amos, R., and Brewer, P. (1972) Practical Liquid Chromatography, New York, London.
Belyaevskaya. T. A., Bol'shova, T. A., and Brykina, G. D. (1975) Chromatography of Inorganic Substances, Van Nostrand Reinhold, New York. DOI: 10.1016/S0021-9673(00)87864-4
Heftmann, E. (ed.) (1975) Chromatography, a Laboratory Handbook of Chromatographic and Electrophoretic Methods, Van Nostrand Reinhold, New York.
References
- Perry, S., Amos, R., and Brewer, P. (1972) Practical Liquid Chromatography, New York, London.
- Belyaevskaya. T. A., Bol'shova, T. A., and Brykina, G. D. (1975) Chromatography of Inorganic Substances, Van Nostrand Reinhold, New York. DOI: 10.1016/S0021-9673(00)87864-4
- Heftmann, E. (ed.) (1975) Chromatography, a Laboratory Handbook of Chromatographic and Electrophoretic Methods, Van Nostrand Reinhold, New York.