This is the emission of light induced by the absorption of light. Two types of emission are usually considered: Fluorescence, which occurs within about one hundred nanoseconds of excitation; and Phosphorescence, which is longer lived.
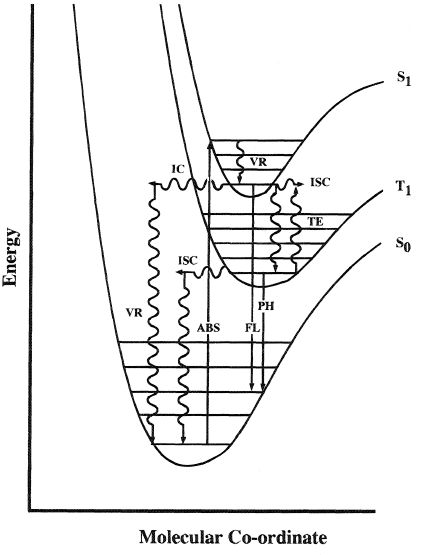
Figure 1 shows the essential features of molecular pbotoluminescence as discussed by Turro (1991). Schematic potential energy curves for the ground state singlet, S0, first excited singlet state, S1, and first triplet state, T1, are shown with associated vibrational energy levels. Transitions between states of the same spin multiplicity are allowed and occur rapidly; those between states of different multiplicity are forbidden and usually occur relatively slowly. Absorption (ABS) populates vibrational levels in S1 and S2. In condensed phases these generally relax within a few picoseconds via vibrational relaxation (VR) and internal conversion (IC) to the lowest vibrational level of S1. At this point the relative rates of intersystem crossing (ISC), internal conversion (IC), and fluorescence (FL) determine the fluorescence and triplet quantum yields. If ISC occurs, then vibrational relaxation gives the lowest vibrational level of T1 which generally decays via reverse ISC to S0 at room temperature or phosphorescence at low temperatures.
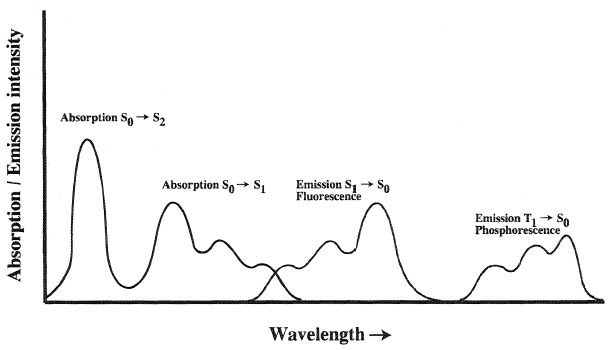
Figure 2 shows the relative positions of absorption and emission bands. It can be seen that the degradation of the excitation energy leads to a Stokes' shift, i.e., fluorescence lies at lower energy than excitation, and that phosphorescence occurs at lower energy than fluorescence. Repopulation of S1, by thermal excitation (TE) from T1 or triplet-triplet annhiliation, can give rise to a weak but long-lived "delayed fluorescence". Organic photoluminescent materials are discussed by Krasovitskii and Bolotin (1988).
The theory and applications of solid state luminescence are discussed by Kitai (1993). The term phosphorescence is often used in solid state photophysics to indicate a temperature dependent emission process involving recombination of trapped electrons and holes (see also Thermoluminescence). Photoluminescence in solids arises from transitions involving conduction bands, valence bands, or localized energy levels at impurity or defect sites. Luminescent centers can be introduced into inorganic crystal or glass matrices by the addition of lanthanide or transition metal ion dopants. Emission and absorption bands may be narrow if the transitions involve only the luminescent center, or broad-band if lattice vibrations are involved. In the latter case large Stokes' shifts may be observed as excitation energy is lost to lattice vibrations before emission. Applications of solid state luminescence include the ruby and neodymium lasers, and phosphors for fluorescent lamps.
Photoluminescence is an extremely sensitive analytical and imaging technique. Laser induced fluorescence from iodine vapor has been used in flow visualization studies by Zimmerman and Miles (1983). The long-lived emission from a suspended ZnS phosphor has been used by Nakatani and Yamadato (1983) to follow the flow of a glycerin/water mixture.
REFERENCES
Kitai.A. H. (1993) Solid State Luminescence Theory Materials and Devices, Chapman and Hall, London.
Krasovitskii, B. M. and Bolotin, B. M. (1988) Organic Luminescent Materials, VCH, Weinheim.
Nakatani, N. and Yamada, Y. (1983) Flow Visualisation III (Ed. W. J. Yang), Hemisphere Publishing Corporation, 82-86.
Turro, N. J. (1991) Modern Molecular Photochemistry, University Science Books, California.
Zimmerman, M. and Miles, R. B. (1983) Flow Visualisation III (Ed. W. J. Yang), Hemisphere Publishing Corporation, 460-462.
References
- Kitai.A. H. (1993) Solid State Luminescence Theory Materials and Devices, Chapman and Hall, London.
- Krasovitskii, B. M. and Bolotin, B. M. (1988) Organic Luminescent Materials, VCH, Weinheim.
- Nakatani, N. and Yamada, Y. (1983) Flow Visualisation III (Ed. W. J. Yang), Hemisphere Publishing Corporation, 82-86.
- Turro, N. J. (1991) Modern Molecular Photochemistry, University Science Books, California.
- Zimmerman, M. and Miles, R. B. (1983) Flow Visualisation III (Ed. W. J. Yang), Hemisphere Publishing Corporation, 460-462.