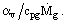A porous medium is a solid with voids distributed more or less uniformly throughout the bulk of the body.
The basic characteristic of this medium is porosity. The bulk porosity Π of a material is defined as the ratio of void volume Vv to body volume V0, Π = Vv/V0. Since the remaining portion Vs of the total volume of the material is in the form of a solid "skeleton", then

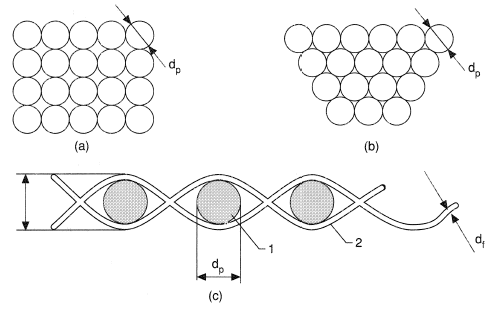
For example, the porosity of porous materials with the skeleton formed by spherical particles with diameter dp can be found from the relation

where Np is the number of particles per unit volume. These spheres can be arranged in various ways (Figures 1a and b). The cubic arrangement of spheres of the same diameter is characterized by a porosity of 0.476, while at a denser, rhombic, packing the porosity reduces to 0.259 (theoretically, this is the minimum porosity of packing of uniform spheres without deformation of the solid). The real porosity generally is estimated using its relation to density ρΣ = ρ0(1−Π) or Π = 1(ρΣ/ρ0), where ρΣ and ρ0 are the densities of the medium and of the solid material forming its skeleton, respectively.
Permeability (or gas permeability) is the property which gives a measure of the gas flow through a porous medium exposed to a pressure difference. The superficial velocity V of fluid flow depends on permeability and pressure gradient in accordance with a modified Darcy equation

Here, superficial velocity v is defined as the volumetric flow rate of the fluid per unit cross section of the medium. The coefficent α allows for friction losses that are characterized by the fluid viscosity η and the structure of a porous matrix. The coefficient of inertia β takes into account the losses associated with expansion, constriction, and bends in the pore channels; these losses are approximately proportional to ρV2.
The modified Darcy equation is universal and describes isothermal liquid and gas flow in any porous solids without allowance for capillary forces. The influence of capillary forces (an increase in viscosity) is observed in water at the pore sizes dp < 1 μm. As a consequence, the ratio of water to gas flow rates for identical pressure difference may be either proportional to the ratio of their viscosities at high dp or decrease by nearly a factor of 20 for porous channels of small diameter.
At low coolant velocity, the inertia term in the Darcy equation may be neglected, and the equation takes the form that is widely used in the theory of filtration

where K = 1/α is the Darcy permeability coefficient.
In this flow regime, the superficial velocity V or superficial mass flux  in porous material is proportional to pressure gradient. The higher the velocity, the stronger the influence of the quadratic term in the resistance law, while the dependence of flow rate on pressure gradient dp/dy gradually weakens and tends to a square root function
in porous material is proportional to pressure gradient. The higher the velocity, the stronger the influence of the quadratic term in the resistance law, while the dependence of flow rate on pressure gradient dp/dy gradually weakens and tends to a square root function

Let us discuss in more detail a viscosity-inertia regime of filtration of a compressible ideal gas. Substituting gas mass flux for filtration velocity V

we transform the modified Darcy equation to the form

If the pressure at the inlet to a porous plate is p1 and at the outlet p2, the temperature being constant throughout its thickness δ, then the solution to this equation is
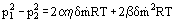
that, after simple manipulations, takes the form

where the Re number is calculated using the characteristic dimension of the porous medium determined as a ratio of the inertial to viscous resistance coefficients β/α
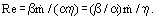
In contrast to the flow in tubes, the choice of characteristic parameters for a porous wall is ambiguous. Even for powdered materials produced by sintering particles of the same shape and size determination of the pore channel diameter as a characteristic dimension involves difficulties and leads to a great scatter of experimental data being processed.
The authors of some papers make an attempt to relate porosity Π with the coefficients α and β in the modified Darcy law. In particular, a dependence of the type of
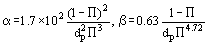
is established for materials produced by sintering of spherical particles with diameter dp, m.
Real materials virtually all have a distribution of grain size, which appreciably reduces porosity and pore size. For instance, the diameter of a pore formed by four spheres with diameter dp is 0.156 dp, by three dp spheres and one 0.5 dp sphere it is as low as 0.118 dp. If, however, another 0.5 dp sphere appears among the four dp spheres, then the pore size reduces to 0.095 dp. Porosity diminishes especially sharply in cases when fine particles are in pores formed by coarse grains.
To secure an adequate mechanical strength, porous materials are subjected to a special thermal treatment, i.e., sintering, at a temperature of the order of 0.8-0.9 of the melting point. The grains of powder, as a rule, possess an inherent residual porosity from 8 to 15 per cent. The overal porosity of metal porous bodies varies over the range 30 to 40 per cent.
Besides pure metals, various carbides, ceramics, and some other materials are used for production of porous bodies. In this case the porous structure is formed by adding an evaporable component to the material and subsequent firing the mixture. This method makes possible the production of materials with porosity up to 50-60 per cent.
Mesh porous matrices can be obtained from fibrous substances (Figure 1c). These materials possess some valuable advantages over powders, viz. a considerably higher strength, easy manufacture of materials with preset and uniform porosity depending on the type of original mesh, and, finally, the possibility of making products of any size.
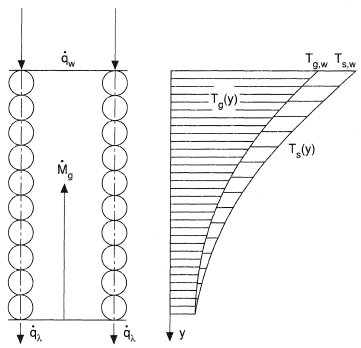
The rate of intraporous convective heat transfer is commonly characterized by the volume heat transfer coefficient αv. If we denote by Ts the local temperature of a porous wall and by Tg the coolant temperature at the same section (Figure 2), then the thermal state of a plane porous wall with constant thermal properties of the material and the coolant will be described in the steady state case by two equations, one
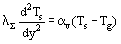
for the solid phase (the porous matrix) and the other

for the (gaseous) coolant.
We introduce a dimensionless temperature difference θ =  , which accounts for a local normalized to the maximum temperature difference across the porous wall. In this case the set of two equations reduces to one equation with respect to θ
, which accounts for a local normalized to the maximum temperature difference across the porous wall. In this case the set of two equations reduces to one equation with respect to θ
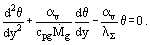
Its general solution is θ = C1ek1y + C2ek2y, where the exponents k1 and k2 are the roots of the characteristic equation

The constants C1 and C2 are determined from the boundary condition on the inner surface and the heat balance in the porous wall. Here, however, there arises an uncertainty in the allowance for the fraction of heat energy, which is supplied to the coolant before entering the porous wall. For instance, flowing along the inner surface of the wall, the coolant may be heated as if in a channel with suction through one of the channel surfaces.
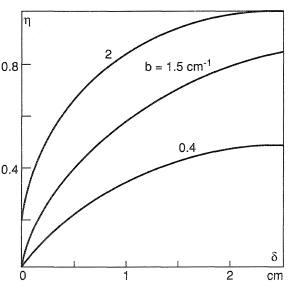
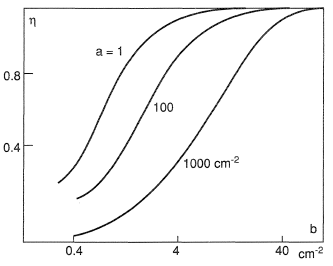
Nevertheless, qualitative characteristics of coolant heating in a porous wall can be specified without elucidating the type of the boundary condition. Figures 3 and 4 demonstrate how two reference parameters entering the equation for θ, namely, a = αv/λΣ and  , affect the efficiency of a porous system η = (1− θw). Figure 3 shows the effect of the porous wall thickness δ at a fixed a = 0.3 cm−2. Figure 4 graphs the dependence of the dimensionless temperature on the set of parameters a and b for a wall thickness δ = 0.6 cm. For a given value of b it is advisable to diminish the coefficient a by, for example, increasing the thermal conductivity of the solid phase, which brings about a flatter temperature profile. The coolant temperature at the outlet from the porous wall may turn out to lower than the temperature of the porous skeleton.
, affect the efficiency of a porous system η = (1− θw). Figure 3 shows the effect of the porous wall thickness δ at a fixed a = 0.3 cm−2. Figure 4 graphs the dependence of the dimensionless temperature on the set of parameters a and b for a wall thickness δ = 0.6 cm. For a given value of b it is advisable to diminish the coefficient a by, for example, increasing the thermal conductivity of the solid phase, which brings about a flatter temperature profile. The coolant temperature at the outlet from the porous wall may turn out to lower than the temperature of the porous skeleton.
Temperature equilibrium in a porous body strongly depends on the thermal conductivity of the porous material λΣ and on the internal heat transfer αv. In porous metals, thermal resistance to heat transfer is mainly observed in the region of contact of neighboring particles (Figure 2), where the smallest cross section area and the greatest inhomogeneity of a metal composition are observed. The quality of thermal contact depends on many practically irreproducible technological factors, including the shape and size of the original particles, material purity and composition, compacting pressure, sintering temperature and time. It is this irresponducibility that excludes accurate physical modeling of heat transfer and accounts for the spread of experimental data.
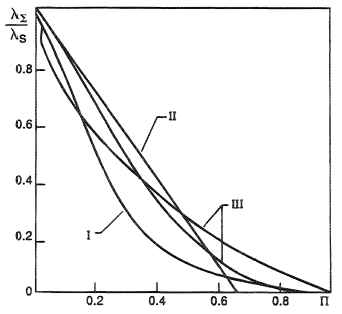
A large number of relationships have been developed for calculating the dependence of the thermal conductivity λΣ on porosity. However, none of them can describe the vast range of experimental data. Therefore, we confine ourselves to two approximating dependences of λΣ on porosity Π denoted by Roman numerals I and II in Figure 5. The great bulk of experimental data denoted by III in Figure 5 lies between these two approximations, and they can be considered an upper and lower estimate of the thermal conductivity coefficient of real porous materials.
Curve I corresponds to a quadratic dependence

Curve II represents the simplest linear function

whose range of applicability is limited by the value Π < 0.6. As is seen from Figure 5, the discrepancy between curves I and II is often more than a factor of two. Other linear approximations, differing from II only by a numerical coefficient of porosity Π, that can vary from 2.1 to 1.5, are also encountered in the literature. It should be pointed out that λΣ measured for fibrous (mesh) and powdered (sintered from spheres) materials differs from the above expressions.
The effect of temperature on thermal conductivity of porous materials is ambiguous. For the materials with a high thermal conductivity of the solid phase (copper, tungsten) the dependence of λΣ on T is of the same character as for the relevant nonporous materials (see Thermal Conductivity). This is indicative of the absence of significant changes in the porous structure and the small contribution of radiant and convective components of heat transfer.
However, the effect of radiation or the kind of coolant gas may play an essential role for ceramic materials characterized by a low thermal conductivity of the matrix (λΣ < 1 W/mK) and a high porosity.
As for heat transfer in a fluid, we must take into account not only the molecular component λgΠ but also the possibility of a convective contribution, which may substantially increase with increase pressure.
For fluid flow in pore channels it is not possible accurately to determine the heat transfer surface. Therefore, we use in this case not the traditional heat transfer coefficient per unit area of interface but the volume coefficient of heat transfer  averaged over the entire volume of the porous medium with an average temperature difference ΔT.
averaged over the entire volume of the porous medium with an average temperature difference ΔT.
Under stationary conditions the quantity of heat  supplied to a coolant is calculated by its heating in a porous wall
supplied to a coolant is calculated by its heating in a porous wall

or by the specific internal heat release as a result of ohmic heating

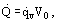 being totally determined by the current and the electrical resistance of a porous matrix. The main difficulty in experiments is measuring temperature difference ΔT = Tgw − Tg0. The temperature difference cannot in actual fact be measured; therefore, the porous wall temperature is measured and the gas temperature obtained by heat balance.
being totally determined by the current and the electrical resistance of a porous matrix. The main difficulty in experiments is measuring temperature difference ΔT = Tgw − Tg0. The temperature difference cannot in actual fact be measured; therefore, the porous wall temperature is measured and the gas temperature obtained by heat balance.
Uncertainty in αv is also introduced by errors in temperature measurements in the porous medium. Apart from common errors due to heat removal thermocouple junctions, there arise errors brought about by jamming of pores by the thermocouple, intense localized heat transfer between the coolant and the thermocouple, nonuniform heating of the thermocouple as a result of radiation from the matrix itself or from an external source under radiant heating. All the above factors are responsible for a substantial scatter of experimental data.
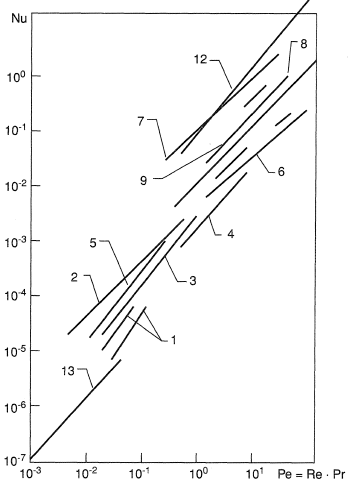
The results of experimental determination of the volume heat transfer coefficient in porous media are summarized in Figure 6 (the numerals from 1 to 13 refer to the published papers). The comparison of the data obtained is hampered by different choice of characteristic dimensions for the Re and Nu numbers. In contrast to the surface heat transfer, the Nu number involves a squared characteristic linear dimension Nu = αvl2/λ . Some researchers use as l the diameter of original particles dp, others prefer the mean pore diameter, but most believe it more correct to take the ratio of coefficients of viscous α to inertial β resistance in the modified Darcy law.
The merit of the latter technique for the characteristic dimension choice is the fact that porosity Π is excluded from the similarity equation Nu = f(Re). It follows from the comparison of the experimental data for various coolants such as air, alcohol and transformer oil that their individual properties can be taken into account by introducing the Pr number to the same extent as by introducing the Re number. The product Re Pr = Pe is the Peclet number.
The data in Figure 6 make it possible to select various approximation dependences of the Nu = A Pen type, where the exponent n on the Pe number in the papers mentioned varies from 0.65 to 1.84. Hence, any averaged empirical relation can be taken only as a first approximation. Choosing the β/α ratio as a characteristic dimension enables us to write the similarity relation for internal heat transfer as
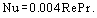
Account must also be taken of a limited thickness of the porous wall. The experimental results become independant of layer thickness only after 8 to 10 layers of spheres have been laid. However, the results are relatively insensitive to random fluctuations of the coolant flow rate  .
.
REFERENCES
Gnielinski, V. (1983) Fixed beds. Chapters 2.5.4 of Heat Exhanger Design Handbook, Hemisphere Publishing Corporation, New York.
Les références
- Gnielinski, V. (1983) Fixed beds. Chapters 2.5.4 of Heat Exhanger Design Handbook, Hemisphere Publishing Corporation, New York.