When two or more pure gases, held at a temperature T and a pressure p, are mixed to form a homogeneous gas mixture at the same temperature and total pressure, the relationship between the volume of the mixture and the volume of the pure components is, generally, complicated. However, if the pure components and the mixture conform to the perfect gas equation of state then the volume of the mixture is simply the sum of the volumes of the pure components [Bett et al. (1975)] so that
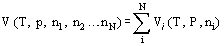
This result is known as Amagat's Law of Additive Volumes. While it is strictly valid only for the mixing of perfect gases, the law is often a useful approximation for real gases.
REFERENCES
Bett, K. E., Rowlinson, J. S., and Saville, G. (1975) Thermodynamics for Chemical Engineers, Athlone Press, London.
Referencias
- Bett, K. E., Rowlinson, J. S., and Saville, G. (1975) Thermodynamics for Chemical Engineers, Athlone Press, London.