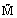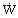|
Absorptivity (radiation)
|
α
|
—
|
|
Amount of substance
|
N
|
mol
|
|
Molar flow rate
| 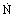 |
mol/s
|
|
Molar mass velocity(= N/Ac)
| 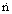 |
mol/m2s
|
|
Angle
|
|
|
|
Plane
|
α, β, γ, θ, Ø
|
rad
|
|
Solid
|
ω
|
sr
|
|
Area
|
|
|
|
Cross-sectional
|
S
|
m2 |
|
Surface
|
A
|
m2 |
|
Coefficient of cubic expansion
|
|
|
|
(1/v)(∂v/∂T)p |
β
|
K−1 |
|
Concentration
|
|
|
|
Mass (= M/V)
|
c
|
kg/m3 |
|
Molar (= N/V)
|  |
mol/m3 |
|
Contact angle
|
θ
|
rad.
|
|
Density
|
|
|
|
Mass (= M/V)
|
ρ
|
kg/m3 |
|
Molar (= N/V)
| 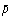 |
mol/m3 |
|
Diameter
|
D
|
m
|
|
Diffusion coefficient
|
D
|
m2/s
|
|
Diffusivity
|
δ
|
m2/s
|
|
Thermal (= λ/ρcp)
|
κ
|
m2/s
|
|
Dryness fraction (quality)
|
x
|
—
|
|
Emissivity (radiation)
|
ε
|
—
|
|
Energy
|
E
|
J = kg m2/s2 |
|
Enthalpy (= U + pV)
|
H
|
J = kg m2/s2 |
|
Specific, molar
|
h,
 |
J/kg, J/mol
|
|
Of phase change at constant p:
|
|
|
|
Melting
|
hsl,
 |
J/kg, J/mol
|
|
Sublimation
|
hsg,
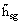 |
J/kg, J/mol
|
|
Evaporation
|
hlg,
 |
J/kg, J/mol
|
|
Entropy
|
S
|
J/K = kg m2/s2K
|
|
Specific, molar
|
s,
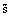 |
J/kg K, J/mol K
|
|
Force
|
F
|
N = kg m/s2 |
|
Fraction
|
|
|
|
Mass, of species i
|
xiyi |
—
|
|
Mole, of species i
|  |
—
|
|
Phase
|
ε
|
—
|
|
Frequency
|
f
|
Hz, s−1 |
|
Frequency circular
|
ω
|
rad/s
|
|
Gas constant
|  |
J/mol K
|
|
Molar (universal)
|
|
= kg m2/s2· mol K
|
|
Specific, of species i
|
Ri |
J/kgK = m2/s2K
|
|
Gravitational acceleration
|
g
|
m/s2 |
|
Gibbs function (H – TS)
|
G
|
J = kg m2/s2 |
|
Specific (h – Ts)
|
g
|
J/kg = m2/s2 |
Molar (
 –
–
 )
)
|  |
J/mol
|
|
|
|
= Kgm2/s2mol
|
|
Heat
|
|
|
|
Quantity of
|
Q
|
J = kg m2/s2 |
|
Rate
| 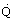 |
W = kg m2/s3 |
Flux (
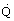 /A)
/A)
|  |
W/m2 = kg/s3 |
|
Heat capacity
|
c
|
J/K = kg m2/s2 K
|
|
Specific, at constant volume or
|
cv, cp |
J/kg K = m2/s2 K
|
|
pressure
|
|
|
|
Molar at constant volume or
|  ,
,
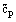 |
J/mol K
|
|
pressure
|
|
= kg m2/s2mol K
|
|
Ratio cp/cv |
γ
|
—
|
|
Heat transfer coefficient
|
α
|
W/m2K = kg/s3K
|
|
Overall
|
U
|
W/m2K = kg/s3K
|
|
Internal energy
|
U
|
J = kg m2/s2 |
|
Specific
|
u
|
J/kg = m2/s2 |
|
Molar
|  |
J/mol = kg m2/s2mol
|
|
Joule Thompson coefficient
|
|
|
|
[(δT/δP)h]
|
μJT |
m2K/N = rns2K/kg
|
|
Length
|
1(L)
|
m
|
|
Diameter
|
d(D)
|
m
|
|
Radius
|
r(R)
|
rn
|
|
Breadth
|
b(B)
|
m
|
|
Height
|
z(Z)
|
m
|
|
Thickness
|
δ
|
m
|
|
Liquid film thickness
|
δ
|
m
|
|
Liquid holdup
|
εL |
—
|
|
Mass
|
M
|
kg
|
|
Flow rate
| 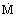 |
kg/s
|
|
Mass velocity or mass flux
|  |
kg/m2s
|
(
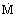 /A)
/A)
|
|
|
|
Flux of species i
|  |
kg/m2s
|
|
Mass transfer coefficient
|
β
|
m/s
|
|
Molar mass
| 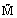 |
kg/kmol
|
|
Pressure
|
p
|
Pa = N/m2 |
|
|
|
= Kg/ms2 |
|
Drop
|
δp
|
Pa = N/m2 |
|
|
|
= kg/ms2 |
|
Reflectivity
|
p
|
—
|
|
Shear stress
|
τ
|
Pa = N/m2 |
|
|
|
= kg/ms2 |
|
Surface tension
|
σ
|
N/m = kgs2 |
|
Temperature
|
|
|
|
Absolute
|
T
|
K
|
|
Difference or interval
|
ΔT
|
K
|
|
Logarithmic mean difference
|
ΔTLM |
K
|
|
Mean temperature difference
|
ΔTM |
K
|
|
Thermal conductivity
|
λ
|
W/m K = kg m/s3K
|
|
Time
|
t
|
s
|
|
Velocity
|
u
|
m/s
|
|
Component in Cartesian
|
|
|
|
coordinates x,y,z
|
u,v,w
|
m/s
|
|
View factor (geometric or
|
Ø12 |
sr
|
|
configuration factor)
|
|
|
|
Viscosity
|
|
|
|
Dynamic (absolute)
|
η
|
Pas = N s/m2 |
|
|
|
= kg/ms
|
|
Kinematic (= η/ρ)
|
ν
|
m2/s
|
|
Void fraction
|
εG |
—
|
|
Volume
|
V
|
m3 |
|
Volumetric flow rate
|  |
m3/s
|
|
Specific, molar volume
|
v,
 |
m3/kg
|
|
Work
|
W
|
J = kg m2/s2 |
|
Rate (power)
| 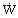 |
W = kg m2/s3 |
|
Wavelength
|
λ
|
m
|
| Subscripts and Superscripts |
|
|
|
Solid or saturated solid
|
S (s)
|
|
|
Liquid or saturated liquid
|
L(l)
|
|
|
Gas or saturated vapor
|
G(g)
|
|
|
Change of phase at constant p:
|
|
|
|
Melting
|
SL (sl)
|
|
|
Sublimation
|
SG (sg)
|
|
|
Evaporation
|
LG (lg)
|
|
|
Critical state
|
c
|
|
|
Initial
|
o
|
|
|
Inlet
|
in,
|
|
|
Outlet
|
out,
|
|
|
At constant value of property
|
p,v,t. etc
|
|
|
Mean
|
–
|
|
|
Molar (per unit of amount of
|
~
|
|
|
substance)
|
|
|
|
Stagnation
|
o
|
|
|
Wall
|
W(w)
|
|

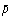


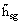

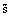


 –
–
 )
)

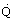
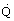 /A)
/A)

 ,
,
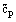

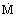

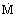 /A)
/A)