Neutrons, together with protons, form the fundamental constituents of atomic nuclei. Neutrons have a mass of 1.675 × 10−27 kg, just slightly greater than that of protons (1.673 × 10−27 kg). Neutrons have a zero electric charge, whereas protons have an electric charge equal in magnitude to that of an electron. Neutrons are found in the nuclei of all atoms with the exception only of hydrogen whose nucleus normally consists of one proton. A few atoms of hydrogen contain nuclei with one proton and one neutron and even fewer have one proton with two neutrons. These rarer forms of hydrogen are known as deuterium and tritium and are called isotopes of hydrogen. Within stable nuclei, such as deuterium, the neutron remains stable indefinitely. Outside the nucleus, however, free neutrons are found to be unstable and decay by beta decay. In this process the neutron changes into a proton and the small reduction in mass produces a high energy electron (beta particle) and an antineutrino (an uncharged and essentially massless particle).
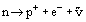
The half-life of free neutrons is approximately 10 minutes. This means that, if at some initial time there are N free neutrons, there will be N/2 free neutrons after 10 minutes and N/2 neutrons will have decayed to protons in that time.
Having no electric charge, neutrons do not cause ionization as they pass through matter. Neutrons, however, interact readily with atomic nuclei since there is no coulomb barrier to overcome. For low energy neutrons, the most likely interaction is radiative capture. In this process, the capture of a neutron by a nucleus is followed by the emission of gamma rays resulting from the conversion of the neutron's binding energy to excitation energy of the compound nucleus. In most nuclei this amounts to about 6–8 MeV. The probability of radiative capture occurring increases, in most nuclei, as the neutron energy decreases in the range below ~0.1 eV. At higher energies, the capture probability (or cross-section) varies in a more complex way, exhibiting a resonance structure. At energies in the 100 keV — 1 MeV range, the neutrons have sufficient energy to eject other particles such as protons or alpha particles from nuclei.
Elastic scattering reactions are those where the neutron shares its kinetic energy with a nucleus, conserving the total kinetic energy. Inelastic scattering occurs with high energy neutrons when part of their kinetic energy is transferred to excitation energy of the nucleus; kinetic energy is then not conserved.
An important neutron-induced reaction is nuclear fission. This occurs in very heavy nuclei such as 235U when the capture of a neutron leads to an excited compound nucleus in which the balance between the Coulomb forces and the strong nuclear forces is such that fission of the nucleus is the most likely outcome. The fission process itself releases further neutrons (about 2.5 per fission) leading to the possibility of a self-sustaining chain reaction. The neutron thus plays a central role in the production of nuclear power and the subject of nuclear reactor physics is concerned mainly with the behavior of neutrons and their interactions with matter. Nuclear Reactors are controlled by adjusting the positions of neutron absorbers within the reactor core.