Gases such as nitrogen, oxygen and methane require the use of very low temperatures to liquefy and store them at relatively low pressures. To achieve this, a whole range of cryogenic technologies has been developed to ensure the economical liquefaction of gases. The reason why such technology has become important is that storage of these gases is more economic and versatile in their liquid form.
There are several ways in which refrigeration can be supplied to a process to cool and/or condense a gas or mixture of gases. These can be grouped into the three fundamental principles used in commercial applications:
The Joule-Thomson effect
Compression/condensation and expansion of a pure component
Expansion turbines or engines.
Most processes in cryogenic technology use one or more of the above principles. Alternatively, they may use a mixed refrigerant as working fluid.
The refrigeration requirements for each cryogenic process are made up of the following elements:
heat ingress from the atmosphere
irreversible thermodynamic losses through heat exchange
the requirement to produce liquid from what is normally a gas.
Heat ingress is usually kept to a minimum by good insulation practices and is a small proportion of the refrigeration requirement in industrial liquefaction processes. Heat exchanger warm-end losses and other irreversible losses are minimized through the use of compact, high-efficiency heat exchangers. Such exchangers are plate-fin aluminum or stainless steel units, as well as wound coil or etched exchangers made in stainless steel or sometimes aluminum. They have economical temperature difference approaches of 2−3°C in contrast with normal shell and tube exchangers, which are economical at much higher temperature differences. (See also Heat Exchangers.)
The importance of good heat transfer in cryogenic processes cannot be overemphasized because of the significant cost of liquefaction of fluids, such as N2, O2 and CH4. The references attached to this section give a more detailed analysis of the benefits of compact heat exchange.
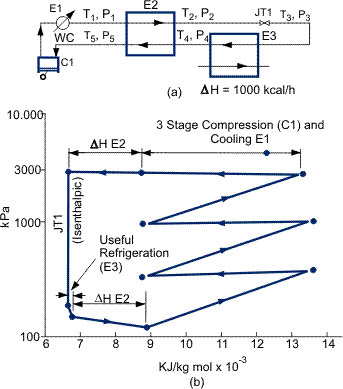
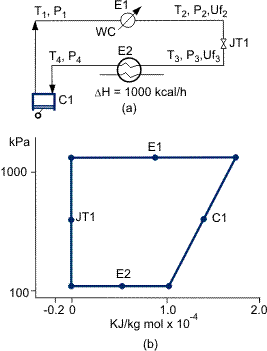
The three basic methods of Refrigeration, used singly or in combination with each other, are compared in Figures 1, 2 and 3 where the cycle is represented above a pressure/enthalpy (P-H) chart, which illustrates refrigeration loads that can be produced by each cycle. The cryogenic plant designer must possess the skill to select the best fluid, mixture, technique or combination of techniques to achieve the optimum refrigeration process.

Figure 2. (a) The process scheme and (b) The P-H diagram for a closed-loop, compression-expansion cycle.
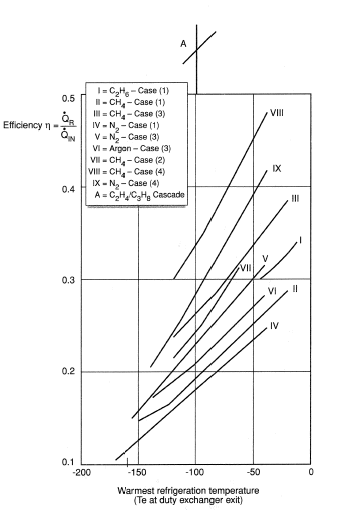
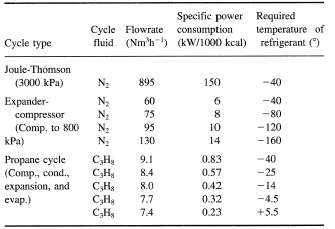
Table 1 compares the different methods from a specific power viewpoint whilst Figure 4 provides a rapid assessment of the overall coefficient of performance of such liquefaction cycles as applied to different temperature levels. This serves as an easy method of estimating the approximate power input for such refrigeration cycles, where  R is the refrigeration duty and
R is the refrigeration duty and 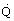 IN is the power input.
IN is the power input.
Table 1. Comparison of three refrigeration cycles. The basis is to provide 1000 kcal h—1 refrigeration at different levels.
REFERENCES
Barron, R. F. (1985) Cryogenic Systems (2nd edn.) Oxford University Press.
Hands, B. A. (1986) Cryogenic Engineering. Academic Press. London.
Haselden, G. G. (1971) Cryogenic Fundamentals, Academic Press, London.
Isalski, W. H. (1989) Separation of Gases, Oxford Science Publications.
Isalski. W. H. (1993) Kempe's Engineers Year Book, Refrigeration B5/1, Benn.
Newton, C. L. (1976) Hydrogen Production and Liquefaction, Chem. & Proc. Eng., (Dec), p. 51.
Scott, R. B. Denton. W. H., and Nicholls, C.M. (1964) Technology and Uses of Liquid Hydrogen, Pergamon Press. Oxford. DOI: 10.1016/0042-207X(64)92181-5
Tomlinson, T. R. et al (1990) Exergy analysis in process development, Chem. Eng., 11 & 25 October, p. 25.
References
- Barron, R. F. (1985) Cryogenic Systems (2nd edn.) Oxford University Press.
- Hands, B. A. (1986) Cryogenic Engineering. Academic Press. London.
- Haselden, G. G. (1971) Cryogenic Fundamentals, Academic Press, London.
- Isalski, W. H. (1989) Separation of Gases, Oxford Science Publications.
- Isalski. W. H. (1993) Kempe's Engineers Year Book, Refrigeration B5/1, Benn.
- Newton, C. L. (1976) Hydrogen Production and Liquefaction, Chem. & Proc. Eng., (Dec), p. 51.
- Scott, R. B. Denton. W. H., and Nicholls, C.M. (1964) Technology and Uses of Liquid Hydrogen, Pergamon Press. Oxford. DOI: 10.1016/0042-207X(64)92181-5
- Tomlinson, T. R. et al (1990) Exergy analysis in process development, Chem. Eng., 11 & 25 October, p. 25.