In heat storage, use is made of the thermal capacity of solid or liquid materials, either by their sensible (specific) heat effect (heating/cooling cycles) or by their latent heat effect at a phase change (melting/freezing cycles). For heat storage, the important thermal characteristics are:
Heat capacity. MJ/m3
Charge and discharge rates,
 CP (Ti − T0), kW/m3
CP (Ti − T0), kW/m3Heat loss rate
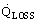 , kW/m3
, kW/m3Thermal stratification, ΔTSTR
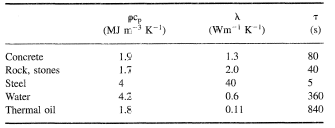
When use is made of the sensible heat effect a high volumetric specific heat (ρcp) of the material is required. Table 1 gives some data for typical materials. Solid materials are often used in rock (packed) beds. As packing densities are often between 60% and 85% the volumetric capacity (ρcp) is reduced proportionally. For higher temperatures the use of such packed beds as regenerators is common. For low temperature applications like home heating and solar energy, water storage is mostly used.
In latent heat stores the latent heat effect is important. At the phase change temperature the heat of solidification will be available when freezing a liquid. Table 2 gives some latent heat effects and the solidification temperature Ts for several materials. For some organic materials, there is a transition range ΔTtr . Inorganic materials may show supercooling effects, which means that the heat discharges at a (unwanted) lower temperature. Also segregation can occur in inorganic solutions in water.
To load/unload a heat storage vessel a heat transfer fluid (like water, air, oil) takes up the heat either by direct contact or through a heat exchanger. Often the limiting heat transfer is on the heat storage material side. Conductivity (λ) in solid materials limits the heat transfer rate. In Table 1 a typical discharge time τ based on transient heat conduction problem for a Fourier number Fo = æτ/d2 equal to 0.5 has been given for a typical dimension d of 10 mm (see Fourier Number). The time τ gives the time for a 90% or more completed loading/unloading cycle. It increases proportional to d2. Also for the solidification (unloading) part of a latent heat store this is the limiting heat transfer mechanism. For liquid heat stores and in melting, natural convection effects will increase the heat transfer rate. For organic latent heat materials with low conductivity-values finned pipes and embedded metal structures are used to improve the heat transfer.
Heat losses of the storage vessel to the surroundings can severely diminish the heat storage efficiency.
In sensible heat stores the heat is loaded/unloaded in an axial direction. In this direction, a temperature gradient (ΔTSTR) will occur. In the optimal case, there is a rather sharp temperature front. However, due to axial dispersion of heat, and in liquids also due to convection flows, this front will be spread out. With large convection flows it can even completely disappear and result in a single, mixed temperature. Due to the dispersion the average temperature in the storage at the end of the loading will decrease. This lowers the storage efficiency.
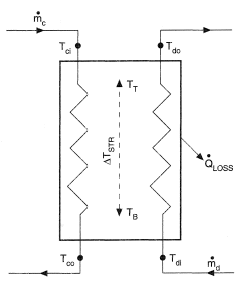
Figure 1. Principles of heat storage. Heat charge rate: 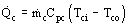 , heat discharge rate:
, heat discharge rate: 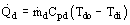 , temperature in top of store TT and bottom of store TB.
, temperature in top of store TT and bottom of store TB.
REFERENCES
Hoogendoorn, C. J. and Bart, G. C. J. (1992) Performance and modelling of lat. heat stores, Solar Energy, 48, 1, 53-58.
Ouden, C. den (1981) Thermal Storage of Solar Energy, Martinus Nijhoff Publishers, the Hague, NL.
Paykoc, E. and Kakac, S. (1987) Solar Thermal Energy Storage; in Solar Energy Utilization, H. Yüncü , Ed., NATO ASI Series, Martinus Nijhoff Publishers, Dordr., NL.
References
- Hoogendoorn, C. J. and Bart, G. C. J. (1992) Performance and modelling of lat. heat stores, Solar Energy, 48, 1, 53-58.
- Ouden, C. den (1981) Thermal Storage of Solar Energy, Martinus Nijhoff Publishers, the Hague, NL.
- Paykoc, E. and Kakac, S. (1987) Solar Thermal Energy Storage; in Solar Energy Utilization, H. Yüncü , Ed., NATO ASI Series, Martinus Nijhoff Publishers, Dordr., NL.