Flotation is a process in Liquid-Solid Separation technology whereby solids in suspension are recovered by their attachment to gas (usually air) bubbles, usually with objective of removing the solids from the liquid. The particles most effectively removed are in the size range from 10 to 200 μm. The particle-bubble aggregates that are formed have a density less than the suspension itself; they rise to the surface and are removed. In the minerals industry, selective froth flotation is used to concentrate specific species of particles in a finely ground ore. "Flotation" is a generic term for a number of processes known as "adsorptive bubble techniques", Lemlich (1972); these are classified into foaming and nonfoaming separation methods. Foaming methods require the addition of surfactants to generate a relatively stable foam or froth which then acts as a carrier fluid during particle removal. The properties most important in determining the success of a flotation process are solid hydrophobicity, bubble to particle size ratio, and the extent of turbulence in the fluid.
Most inorganic, and many organic, particles have hydrophilic surfaces, and as such they are not floatable. To attach a gas bubble to these solids therefore requires the displacement of a water film from their surfaces. This is achieved using surfactants (known as collectors) to render the particle surface hydrophobic. Collectors are usually long chain hydrocarbon molecules containing polar groups; these adsorb on to the surface at the charged group, with the hydrocarbon chain being presented to the aqueous phase. (See Surface Active Substances.)
Other reagents used in flotation are frothers to promote a smaller bubble size and a more stable froth, and modifiers or conditioners to adjust the pH of the suspension and assist attachment of bubbles to specific particle species in the suspension. Conditioners used include coagulants and polymer flocculants.
All flotation systems require a source of gas bubbles. In natural flotation, release of gas in the form of bubbles from, for example, a fermentation is the cause of scum at the surface of the liquid. Aided flotation is a natural flotation improved by blowing air bubbles into the suspension, and is useful for the separation of greases dispersed in a turbid liquid. In most process industry flotations, the bubbles are formed by three main techniques: (i) mechanically, using an agitator combined with an air injection system or by pumping the air through a porous plate or nozzle, to form bubbles that measure 0.2 to 2 mm in diameter (mechanical flotation or froth flotation); (ii) by gas nucleation from a solution to form bubbles of about 40 to 70 mm in diameter, induced by subjecting the suspension to a vacuum (vacuum flotation), or by saturating water with the gas under pressure and injecting it into the suspension (dissolved air flotation), or by supersaturating the suspension under pressure and then relieving the pressure (microflotation); and, (iii) by electrolysis of the aqueous phase (electroflotation) to produce bubbles < 50 μm with minimal turbulence.
As general purpose liquid-solid separation technologies, the two flotation techniques which have found increasing value in recent years are dissolved air (DAF) and electroflotation (EF). EF has the advantage that no pressurized recycle is required; the flow handled by the EP unit comprises the feed only. The recycle employed with DAE increases the flow into the unit. In EF there is no need for recycle pumps, compressors or saturators, but EF does require transformer-rectifier systems which can be relatively expensive. Both DAF and EF may be used either for clarification of liquids or for thickening or concentrating of solids suspensions. Power consumptions and air flows are dependent on many factors, but comparable data (per m3 of water treated) may be given for water treatment applications: aided flotation (grease removal) requires about 5 to 10Wh m−3 and 100 to 400 Nl m−3, froth flotation 60 to 120 Wh m−3 and 10,000 Nl m−3, and DAF (clarification) 40 to 80 Wh m−3 and 15 to 50 Nl m−3.
The microscopic mechanisms that occur in flotation are complex, making it impossible to describe in mathematical detail or to develop fundamental design equations. According to Fuerstenau (1976), most flotation systems with a high degree of turbulence approximate to perfect mixers and the recovery of solids is then given by:
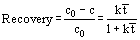
In those dissolved air flotation systems where turbulence is low and in electroflotation cells, hydrodynamic conditions are probably closer to plug flow. In practice, it is difficult to achieve plug flow conditions, and a reasonable approximation to describe recovery by flotation is:

In these equations, c is the solids concentration, c0 is the solids concentration in the feed, and
 is the mean retention time of the suspension. Comparing the above equations shows that theoretically a plug flow flotation cell is more efficient than a perfectly mixed system (for the same k and
is the mean retention time of the suspension. Comparing the above equations shows that theoretically a plug flow flotation cell is more efficient than a perfectly mixed system (for the same k and
 ); to put this another way, a smaller plug flow tank will give the same recovery as a larger perfectly mixed system [Jowett and Sutherland (1979)].
); to put this another way, a smaller plug flow tank will give the same recovery as a larger perfectly mixed system [Jowett and Sutherland (1979)].
Stevenson (1986) has outlined practical methods for the determination of design and scale-up data for flotation systems. (See also Foam Fractionation.)
REFERENCES
Fuerstenau, M. C., (Ed.) (1976) Flotation, A. M. Gaudin Memorial Volume. American Institute of Mining Engineers, New York.
Jowett, A. and Sutherland, D. (1979) A simulation study of the effect of cell size on flotation costs, The Chemical Engineer, 603-607.
Lemlich, R. (1972) Adsorptive Bubble Separation Techniques, Academic Press, London.
Stevenson, D. G. (1986) Chapter 5, Solid/Liquid Separation Equipment Scale-up, (D. B. Purchas and R. J. Wakeman, Eds.). Uplands Press and Filtration Specialists, London.
References
- Fuerstenau, M. C., (Ed.) (1976) Flotation, A. M. Gaudin Memorial Volume. American Institute of Mining Engineers, New York.
- Jowett, A. and Sutherland, D. (1979) A simulation study of the effect of cell size on flotation costs, The Chemical Engineer, 603-607.
- Lemlich, R. (1972) Adsorptive Bubble Separation Techniques, Academic Press, London. DOI: 10.1002/cite.330450427
- Stevenson, D. G. (1986) Chapter 5, Solid/Liquid Separation Equipment Scale-up, (D. B. Purchas and R. J. Wakeman, Eds.). Uplands Press and Filtration Specialists, London.