THE BLACKBODY
1. INTRODUCTION
All objects emit electromagnetic radiation. No intervening medium is required between two locations for radiation exchange to occur, so radiative energy can propagate through a vacuum. Radiative properties of surfaces, particles, and gases usually vary substantially over the wavelength (or frequency) spectrum.
Electromagnetic radiation can be classified according to its wavelength λ0 (where the subscript 0 indicates the value measured in a vacuum), or its frequency ν, where ν = c0/λ0 and c0 is the speed of light in a vacuum. The wave number is also used, especially in spectroscopy, and is defined as η0 = 1/λ0. As electromagnetic radiation propagates from one medium to another, its wavelength changes as a function of the index of refraction, as n = λ0/λ = (c0/ν)/(c /ν). The frequency ν remains the same; for that reason, the use of frequency to describe the spectral behavior of matter is common. The wavelength is usually given in the units of micrometers (μm) or nanometers (nm), where 1 μm is 10-6 m or 10-4 cm, and one nm is 10-9 m. Sometimes, the units are expressed in Angstroms (Å), where 1 μm = 104 Å or 1 nm = 10 Å. The wave number is given in units of cm-1. Visible light is confined to approximately 400-700 nm (0.4-0.7 μm). Thermal radiation is concerned with a wavelength range of 0.1-100 μm, including the ultraviolet and visible (λ = 0.1-0.7 μm), near-infrared (λ = 0.7-10 μm), and far-infrared spectra (λ = 10-100 μm).
Now, consider the definition of the solid angle, used to determine the angular increment containing radiation propagating in a given direction. The solid angle dΩ is determined by dividing the normal base area by the square of the distance from the tip to the base, as shown in Fig. 1. The solid angle dΩ is analogous to the planar angle dθ and its relation to the radius R and arc length dS.
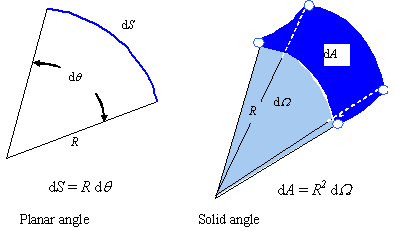
Figure 1. Solid angle, used to determine the angular increment containing radiation propagating in a given direction
Consider an elemental surface area dA surrounded by a hemisphere of radius r. A hemisphere has a surface area 2πr 2 and it subtends a solid angle of 2π steradians (sr) about the center of its base. Hence, for a hemisphere of unit radius of r = 1, the solid angle about the center of the base equals the area on the unit hemisphere. Direction is measured by the zenith and azimuthal angles θ and ϕ where θ is measured from the direction normal to dA. The angular position for ϕ=0 is arbitrary, but is usually measured from the x -axis. A solid angle anywhere above dA is equal to the intercepted area on the unit hemisphere (see Fig. 2). An element of this hemispherical area is given by (sinθdϕ)(dθ) so that
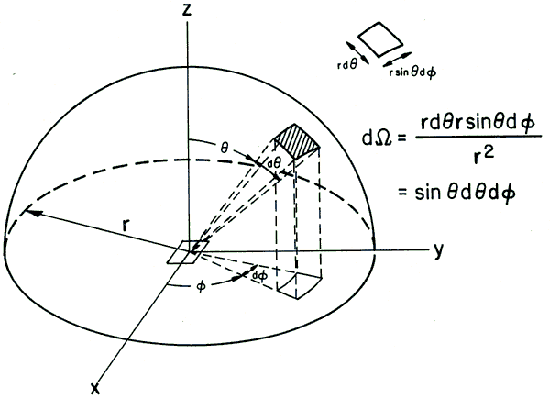
Figure 2. A solid angle anywhere above dA is equal to the intercepted area on the unit hemisphere.
 | (1) |
The integral of dΩ over all angles of a surrounding hemisphere yields 2π sr, units for angular division,
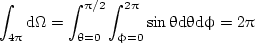 | (2) |
The solid angle is similar to an angle in a plane, but is given in 3D space. The planar angle is the ratio of the chord length to the distance from the apex to the base, while the solid angle is the ratio of base area to the square of the chord length.
2. SPECTRAL RADIATIVE INTENSITY
Consider the spectral radiative energy dQλ(S, Ω,t ) propagating in direction Ω along path S. The spectral radiative intensity Iλ is defined as the spectral radiative energy dQλ per unit projected area dA along a path S, per the solid angle dΩ in steradians around Ω, per unit time interval dt at time t, and a small wavelength interval dλ at around λ,
 | (3) |
The spectral radiative energy is the fundamental quantity for all radiation calculations. The units of Iλ(S, Ω,t ) are Watts/(meter2-micrometer-steradian).
The spectral intensity is always specified at a wavelength λ per unit wavelength interval around that λ. The spectral intensity refers to radiation in an interval dλ around a single wavelength, while the total intensity refers to combined radiation including all wavelengths.
Every material at finite temperature emits radiative energy. Emission from a material is a function of zenith and azimuthal angles, and the local material conditions, including the properties and temperature,
 | (4) |
where T = T (x, y, z ). The body that emits the maximum amount of energy uniformly in all directions and at each wavelength interval is called a blackbody. An ideal blackbody absorbs all radiation incident on it. As a perfect absorber, it serves as a standard with which real materials can be compared.
The quantitative radiative properties of the ideal blackbody have been well established by quantum mechanics and also verified by experiments. Aside from being a perfect absorber for all incident radiation, the blackbody has other important features. Standard texts (Siegel and Howell, 2010; Modest, 2003) show that because the blackbody absorbs the maximum possible radiation at each wavelength and from each direction, it also emits the maximum total amount of radiation, and the blackbody is a perfect emitter at each wavelength. The total radiant energy emitted by a blackbody in vacuum is a function only of its temperature.
The spectral intensity emitted by a blackbody is written as Iλb . The subscripts denote, respectively, that a spectral quantity is being considered and that the properties are for a blackbody,
 | (5) |
The intensity of radiation from a blackbody, defined on the basis of its projected area, is independent of the direction of emission.
The emissive power of a blackbody is the energy per unit time, solid angle, wavelength interval, and area (not projected area), and so is a function of zenith angle θ, and is given by
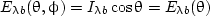 | (6) |
This relation is known as Lambert’s cosine law. Surfaces having a directional emissive power that follows this relation are called Lambertian, diffuse, or cosine law surfaces. A blackbody is always diffuse and serves as a standard for comparison with the directional properties of real surfaces.
The hemispherical spectral emissive power, Eλb , is the energy emitted by a black surface per unit time per unit area and per unit wavelength interval around λ into all directions. The blackbody spectral emission passing though the entire hemisphere, in terms of the blackbody intensity, is
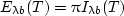 | (7) |
so the blackbody hemispherical emissive power is π times the blackbody intensity.
3. PLANCK'S LAW: SPECTRAL DISTRIBUTION OF EMISSIVE POWER
The spectral emissive power of a backbody is given by quantum theory as
 | (8) |
Here, λ is the wavelength in the medium adjacent to the radiating surface of refractive index n. This is Planck’s spectral distribution of emissive power. For most engineering applications, radiative emission is into air or other gases, for which the index of refraction, n = c0/c, is unity. Planck’s blackbody distribution is expressed in terms of two universal constants, namely, Planck’s constant, h = 6.62606876 × 10-34 J s, and the Boltzmann constant, kB =1.3806503 × 10-23 J/K. Two auxiliary radiation constants are defined as C1 = hc02 and C2= hc0/kB . Alternative forms are in terms of frequency or wave number. In terms of frequency, Eq. (8) becomes
 | (9) |
The quantity Evb (v) is the emissive power per unit frequency interval about v. The intensity is Ivb (v) = Evb (v)/πs so that
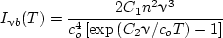 | (10) |
The quantity Eηb (η) is the emissive power per unit wave number interval about η. The intensity is then
 | (11) |
Dividing by T 5 allows casting the blackbody distribution in terms of the single variable λ T,
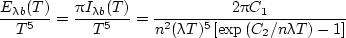 | (12) |
This is shown in Fig. 3.
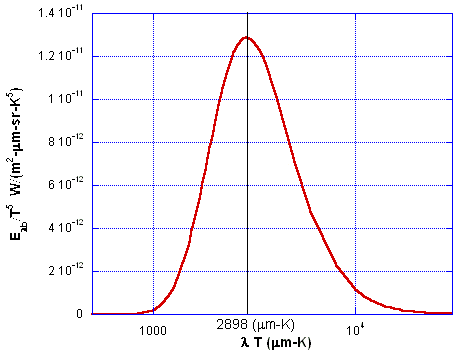
Figure 3. Normalized blackbody spectral emissive power.
4. WIEN'S DISPLACEMENT LAW
The wavelength λmax occurs where the blackbody intensity Iλb (T) is a maximum for a given temperature. This maximum shifts toward shorter wavelengths as the temperature is increased. The value of λmaxT is at the peak of the distribution curve. It is obtained by differentiating Planck’s distribution with respect to λT, and setting the left side equal to zero. If the index of refraction n= 1, this gives
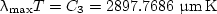 | (13) |
which is Wien’s displacement law.
The total blackbody intensity includes radiation for all wavelengths,
 | (14) |
where the constant σ is
 | (15) |
The hemispherical total emissive power of a blackbody radiating into vacuum is then
 | (16) |
which is the total emitted blackbody energy flux (in watts per meter squared). This is the Stefan-Boltzmann law, and σ is the Stefan-Boltzmann constant.
The fraction of the total emissive power emitted in a wavelength interval, Fλ1T → λ2T , is given by
 | (17) |
The integral can be expressed by two integrals, each with a lower limit λ = 0, so that
 | (18) |
The fraction of the emissive power for any wavelength interval can therefore be found by having F0 → λ1T as a function of λ.
The F function can be put in terms of the single variable λT as
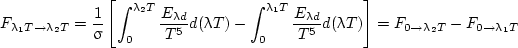 | (19) |
A convenient series form for F0 → λT is found by using the substitution ζ = C2/λT to obtain
 | (20) |
By using the series expansion (e ζ - 1)-1 = e -ζ + e -2ζ + e -3ζ + ··· and then integrating by parts, F0 → λT becomes (Chang and Rhee, 1984)
 | (21) |
where ζ = C2/λT. The infinite series converges very rapidly, and the first three terms of the summation give good results over most of the range of F0 → λT . As λT becomes large, a larger number of terms is required.
5. SUMMARY OF BLACKBODY PROPERTIES
The blackbody posses fundamental properties that make it a standard with which real radiating bodies can be compared. These are as follows:
The blackbody is the best possible absorber and emitter of radiant energy at any wavelength and in any direction.
The total (including all wavelengths) radiant intensity and hemispherical total emissive power of a blackbody into a medium with constant index of refraction n are given by the Stefan-Boltzmann law, πIb = Eb = n 2σT 4.
The blackbody spectral and total intensities are independent of the direction so that emission of energy into a direction θ away from the surface normal direction is proportional to the projected area of the emitting element, dA cosθ. This is known as Lambert’s cosine law.
The blackbody spectral distribution of intensity for emission into a medium with refractive index n is given by Planck’s distribution,

The wavelength at which the maximum spectral intensity of radiation into a medium with refractive index n for a blackbody occurs is given by Wien’s displacement law,
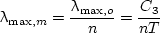 |
REFERENCES
Chang, S. L. and Rhee, K. T., Blackbody Radiation Functions, Int. Comm. Heat Mass Transfer, vol. 11, pp. 451-455, 1984.
Modest, M. M., Radiation Heat Transfer, 2nd ed., Academic Press, New York, 2003.
Siegel, R. and Howell, J. R., Thermal Radiation Heat Transfer, 5th ed., Taylor and Francis, New York, 2010.
References
- Chang, S. L. and Rhee, K. T., Blackbody Radiation Functions, Int. Comm. Heat Mass Transfer, vol. 11, pp. 451-455, 1984.
- Modest, M. M., Radiation Heat Transfer, 2nd ed., Academic Press, New York, 2003.
- Siegel, R. and Howell, J. R., Thermal Radiation Heat Transfer, 5th ed., Taylor and Francis, New York, 2010.