A multiphase flow is defined as one in which more than one phase (i.e., gas, solid and liquid) occurs. Such flows are ubiquitous in industry, examples being gas-liquid flows in evaporators and condensers, gas-liquid-solid flows in chemical reactors, solid-gas flows in pneumatic conveying, etc. This introductory article attempts to give an overview, with more detailed material appearing on each individual type of multiphase flow in separate entries.
In multiphase flows, solid phases are denoted by the subscript S, liquid phases by the subscript L and gas phases by the subscript G. Some of the main characteristics of these three types of phases are as follows:
In a multiphase flow, the solid phase is in the form of lumps or particles which are carried along in the flow. The characteristics of the movement of the solid are strongly dependent on the size of the individual elements and on the motions of the associated fluids. Very small particles follow the fluid motions, whereas larger particles are less responsive.
In a multiphase flow containing a liquid phase, the liquid can be the continuous phase containing dispersed elements of solids (particles), gases (bubbles) or other liquids (drops). The liquid phase can also be discontinuous, as in the form of drops suspended in a gas phase or in another liquid phase. Another important property of liquid phases relates to wettability. When a liquid phase is in contact with a solid phase (such as a channel wall) and is adjacent to another phase which is also in contact with the wall, there exists at the wall a triple interface, and the angle subtended at this interface by the liquid-gas and liquid-solid interface is known as the Contact Angle.
As a fluid, a gas has the same properties as a liquid in its response to forces. However, it has the important additional property of being (in comparison to liquids and solids) highly compressible. Notwithstanding this property, many multiphase flows containing gases can be treated as essentially incompressible, particularly if the pressure is reasonably high and the Mach Number, with respect to the gas phase, is low (e.g., < 0.2).
The most common class of multiphase flows are the Two-Phase Flows, and these include Gas-Liquid Flow, Gas-Solid Flow, Liquid-Liquid Flow and Liquid-Solid Flow. The reader is referred to the general overview article on Two-Phase Flows and to the individual articles on each of the respective two-phase flow types.
Three-phase flows are also of practical significance, and examples are as follows:
Gas-liquid-solid flows: this type of system occurs in two-phase fluidized bed and gas lift chemical reactors where a gas-liquid reaction is promoted by solid catalyst particles suspended in the mixture.
Three-phase, gas-liquid-liquid flows: mixtures of vapors and two immiscible liquid phases are common in chemical engineering plants. Examples are gas-oil-water flows in oil recovery systems and immiscible condensate-vapor flows in steam/hydrocarbon condensing systems.
Solid-liquid-liquid flows: An example here would be that of an immiscible liquid-liquid reaction, in which a solid phase is formed, that separates out in the system.
Multiphase flows are not restricted to only three phases. An example of a four phase flow system would be that of direct-contact freeze crystallization in which, for example, butane liquid is injected into solution from which the crystals are to be formed, and freezing occurs as a result of the evaporation of the liquid butane. In this case, the four phases are, respectively, butane liquid, butane vapor, solute phase and crystalline (solid) phase.
The mean phase content (εi) of the ith phase is defined as the time-averaged volume fraction of that phase in a section of the channel or as the time-averaged area fraction of the phase in a given cross section (the two definitions may be taken as equivalent in most practical situations). Specifically, the mean phase content of the gas phase (εG) is often termed the Void Fraction. The volume flux (or superficial velocity) Ui of a phase is defined as:

where
 is the volume flow rate of the phase (m3/s) and S is the channel cross-sectional area (m2). The total superficial velocity U is given by:
is the volume flow rate of the phase (m3/s) and S is the channel cross-sectional area (m2). The total superficial velocity U is given by:

where n is the total number of phases present. The average phase velocity (ui) of the ith phase is given by:

and the flow quality xi of the ith phase is defined as:

where
 is the mass flux of the ith phase (given by
is the mass flux of the ith phase (given by
 /S, where
/S, where
 is the mass rate of flow the phase through the channel). A multiphase density ρMP may be defined as the mass of the multiphase mixture per unit channel volume, and this is given by:
is the mass rate of flow the phase through the channel). A multiphase density ρMP may be defined as the mass of the multiphase mixture per unit channel volume, and this is given by:

where ρi is the density of the ith phase.
The simplest approach for representation of multiphase flows is to treat them as homogeneous mixtures in which the velocities of all the phases are identical and equal to the homogeneous velocity uH. This is given by:

where
 is the total mass flux,
is the total mass flux,
 the total mass rate of flow and ρH is the homogeneous density given by:
the total mass rate of flow and ρH is the homogeneous density given by:

The homogeneous conservation equations for mass, momentum and energy are stated as follows (detailed derivations are given by Hewitt (1983):
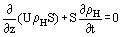


where z is the axial distance, t time, p pressure, g the acceleration due to gravity, α the angle of inclination of the channel, τo the wall shear stress, P the channel periphery,
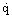 the wall heat flux,
the wall heat flux,
 the internal heat generation rate in the fluid per unit volume and e the energy converted per unit fluid mass, given by:
the internal heat generation rate in the fluid per unit volume and e the energy converted per unit fluid mass, given by:

where h is the specific enthalpy given by:

where μ is the specific internal energy.
For steady state flow in a constant cross-section duct, the momentum equation (Eq. (9)) reduces to:

where the three terms of the right hand are, respectively, the frictional pressure gradient, the accelerational pressure gradient and the gravitational pressure gradient.
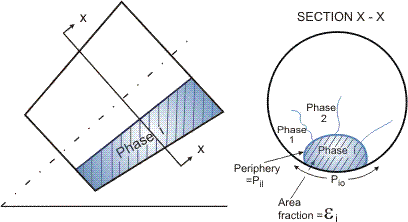
For the separated flow model for multiphase flows, the phases are considered to be flowing in separated zones of the channel, each phase having its own velocity, as illustrated in Figure 1.
To develop equations for this case, it is possible to write the conservation equations for each separate phase, considering the interaction between that phase and the channel wall and also its interactions with the adjacent phases. This approach for two-phase flows is described in detail in the article on Conservation Equations, Two-Phase. A common practice is to add the conservation equations for the respective phases together, and this leads to the mixture conservation equations. Details of the approach for multiphase systems are given by Lahey and Moody (1977) and by Hewitt (1983). Here, for the sake of brevity, we will merely state the mixture conservation equations which, for mass, momentum and energy are respectively:



where hi is the enthalpy of the ith phase, for steady state flow in a constant cross-section duct, Eq. (15) reduces to:
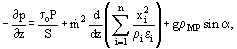
where the three terms on the right hand side of the equation are respectively the factional, accelerational and gravitational pressure gradients.
REFERENCES
Hewitt, G. F. (1983) Multiphase fluid flow and pressure drop: Introduction and fundamentals. Chapter 2.3.1 of the Heat Exchanger Design Handbook, Hemisphere Publishing Corporation, New York.
Lahey, R. T. and Moody, F. J. (1977) The Thermal Hydraulics of a Boiling Water Nuclear Reactor. American Nuclear Society.
References
- Hewitt, G. F. (1983) Multiphase fluid flow and pressure drop: Introduction and fundamentals. Chapter 2.3.1 of the Heat Exchanger Design Handbook, Hemisphere Publishing Corporation, New York.
- Lahey, R. T. and Moody, F. J. (1977) The Thermal Hydraulics of a Boiling Water Nuclear Reactor. American Nuclear Society.