Van der Waals forces are a generic description of the forces that act between atoms and molecules. The fact that liquids and solids are scarcely compressible indicates that when the distances between molecules are small, the forces between them are strongly repulsive. On the other hand, the fact that these condensed phases exist at all indicates that at long range the forces must be attractive.
The force is generally expressed in terms of a potential energy U which, for spherically symmetric molecules, is simply a function of the distance t between two molecules, so that

The general shape of an intermolecular pair potential for a spherically-symmetric system is shown schematically in Figure 1.
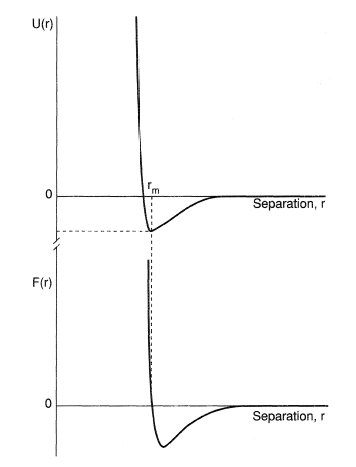
Figure 1. The intermolecular potential and intermolecular force for a pair of spherically-symmetric molecules.
The physical origin of the long-range attractive forces in the case of nonpolar molecules is the interaction between the fluctuating dipoles in each atom caused by the motion of the electrons about the nucleus. The repulsive interactions at short range are caused by the repulsive electrostatic forces between the nuclei of molecules when the electrostatic shielding is reduced as the Pauli Exclusion Principle causes a reduction of the electron density between them. For polyatomic and/or polar molecules, the van der Waals forces also arise as a result of interactions between permanent multipoles within the molecules. The forces and the potential then depend upon the relative orientation of the molecules as well as their separation.
REFERENCES
Maitland, G. C, Rigby, M, Smith, E. B. and Wakeham, W. A. (1961) Intermolecular Forces: Their Origin and Determination, Clarendon Press, Oxford.
Rigby, M, Smith, E. B., Wakeham, W. A. and Maitland, G. C. (1986) An Introduction to Intermolecular Forces, Clarendon Press, Oxford.
References
- Maitland, G. C, Rigby, M, Smith, E. B. and Wakeham, W. A. (1961) Intermolecular Forces: Their Origin and Determination, Clarendon Press, Oxford.
- Rigby, M, Smith, E. B., Wakeham, W. A. and Maitland, G. C. (1986) An Introduction to Intermolecular Forces, Clarendon Press, Oxford.