A particle or droplet will settle in a fluid if its density is greater than that of the fluid in which it is suspended. The (laminar) settling velocity of particles whose concentration is very low, that is when the flow of fluid around a particle does not affect the flow around neighboring particles, can be calculated from Stokes Law:

where D is the diameter of the particle and η is the absolute viscosity of the surrounding fluid. Δρ is the density difference between that of the particle and its surrounding fluid: if Δρ is positive the particle will settle (see Thickeners), or if it is negative the particle will float (see Flotation).
In practice, the concentration of particles in industrial suspensions is usually high enough for there to be significant interactions between particles as they settle (making Stokes Law invalid): these interactions can greatly increase the frictional force at the surfaces of the settling particles. When the effects of mutual interference are negligible, free settling conditions are said to prevail; at higher concentrations hindered settling occurs. The reasons for modification of the settling rate of particles in a concentrated suspension include:
when a wide range of particle sizes are present in the feed, differential settling rates between large and small particles lead to modification of the effective density of the suspension (this is more significant in small scale batch sedimentation),
the upward velocity of the fluid is greater at higher concentrations (causing a decrease in the apparent settling velocity),
the velocity gradients in the fluid surrounding the particles are greater due to the closer proximity of the particles,
the ability of particles to aggregate is enhanced at higher concentrations.
In addition to particle size, density and concentration, and fluid viscosity, other less obvious factors affect the sedimentation rate. These include particle shape and orientation, convection currents in the surrounding fluid, and chemical pretreatment of the feed suspension. Particle with diameters of the order of a few microns settle too slowly for most practical operations; wherever possible these are coagulated or flocculated to increase their effective size, and hence increase their rate of settling.
Sedimentation of a suspension is generally assessed by a jar test, during which a suspension is allowed to settle and the height of the clear liquid (supernatant)-suspension interface is measured as a function of the settling time. In a jar test particles can be observed to settle in any of several quite different ways, dependent on their concentration and their tendency to cohere. The different modes of sedimentation make different demands on the size and shape of a settling tank, and different test procedures are used for evaluating them [details of these are given by Fitch and Stevenson (1986)].
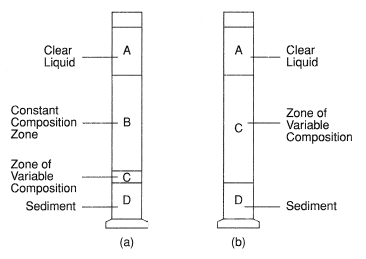
When the particles are, on average, far apart and free to settle individually, clarification sedimentation occurs. This behavior is recognized in a jar test as slower settling particles “string out” behind faster ones, and the supernatant gradually clarifies; solids collect at the bottom of the jar to form a sediment, and the supernatant-suspension interface is generally indistinct.
In more concentrated suspensions, the particles are closer together; in the extreme they can cohere to form a plastic structure which constrains the sedimentation of individual particles. The solids settle with a sharp interface between the pulp and supernatant, and the slurry enters a consolidated or zone settling regime and exhibits zone settling or line settling behavior. These two extremes of behavior are shown in Figure 1.
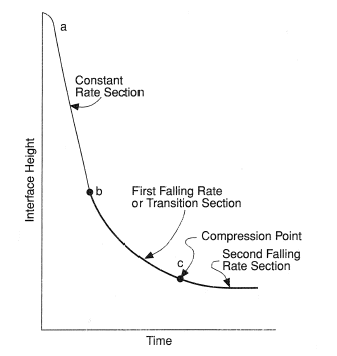
In a jar test exhibiting line settling behavior the interface height can be plotted against time as in Figure 2. After an initial transient the suspension settles at a constant rate in the section from A to B, followed by the first falling rate period from B to C and a second falling rate period from C. The constant rate period corresponds to pulp settling at its initial concentration. During the final falling rate section the pulp is in compression. Point C is known as the compression point or the critical sedimentation point, and identifies the point at which the pulp-supernatant interface goes from zone settling into compression.
Jar test sedimentation data is used as the basis for clarifier or thickener design. Details of design methods can be found in many texts, for example, Fitch and Stevenson (1986) and Osborne (1990). Additional information can be found in Coulson and Richardson (1991) and Rushton (1985). Clarifier design, usually based on "long tube" data, is based on the overflow velocity and the detention time, and calculations are quite simple. Thickener design involves the application of one of a number of alternative models together with jar test data, and is based either on the combined Kynch (1952) and Coe and Clevenger (1916) analysis of settling rates or the Yoshioka (1957) flux analysis. There are many other models, which could be applied, some of which are based on compression of the sediment rather than hindered settling of the suspension. Wilhelm and Naide (1981) recommend the flux analysis method as the most concise and precise for design purposes.
REFERENCES
Coe, H. S. and Clevenger, G. H. (1916) Methods for determining the capacities of slime thickening tanks, Trans AIME, 55, 356, 384.
Coulson, J. M. and Richardson, J. F. (1991) Chapter 5, Chemical Engineering, Vol 2, 4th edn., Pergamon, Oxford.
Fitch, E. B. and Stevenson, D. G. (1986) Chapter 4, Solid/Liquid Separation Equipment Scale-up, D. B. Purchas and R. J. Wakeman, eds., Uplands Press and Filtration Specialists, London.
Kynch, G. J. (1952) A theory of sedimentation, Trans Faraday Soc, 48, 166-176.
Osborne, D. G. (1990) Chapter 5, Solid-Liquid Separation, L. Svarovsky, Ed., Butterworths, London.
Rushton, A., Ed. (1985) Mathematical Models and Design Methods in Solid-Liquid Separation, Nijhoff, Dordrecht.
Wilhelm, J. H. and Naide, Y. (1981) Sizing and operating continuous thickeners, Mining Engineering, 1710—1718.
Yoshioka, N., Hotta, Y, Tanaka, S., Naito, S., and Tongami, S. (1957) Continuous thickening of homogeneous slurries, Chemical Engineering, Tokyo, 21, 66-74.
References
- Coe, H. S. and Clevenger, G. H. (1916) Methods for determining the capacities of slime thickening tanks, Trans AIME, 55, 356, 384.
- Coulson, J. M. and Richardson, J. F. (1991) Chapter 5, Chemical Engineering, Vol 2, 4th edn., Pergamon, Oxford.
- Fitch, E. B. and Stevenson, D. G. (1986) Chapter 4, Solid/Liquid Separation Equipment Scale-up, D. B. Purchas and R. J. Wakeman, eds., Uplands Press and Filtration Specialists, London.
- Kynch, G. J. (1952) A theory of sedimentation, Trans Faraday Soc, 48, 166-176.
- Osborne, D. G. (1990) Chapter 5, Solid-Liquid Separation, L. Svarovsky, Ed., Butterworths, London.
- Rushton, A., Ed. (1985) Mathematical Models and Design Methods in Solid-Liquid Separation, Nijhoff, Dordrecht. DOI: 10.1002/cite.330580626
- Wilhelm, J. H. and Naide, Y. (1981) Sizing and operating continuous thickeners, Mining Engineering, 1710—1718.
- Yoshioka, N., Hotta, Y, Tanaka, S., Naito, S., and Tongami, S. (1957) Continuous thickening of homogeneous slurries, Chemical Engineering, Tokyo, 21, 66-74.