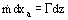Convective flow boiling downstream of the critical heat flux (CHF) location can be termed "post-dryout heat transfer". Conventionally, this regime is divided into a transition boiling region (unstable or partial film boiling) and a stable film boiling region. Transition boiling is defined as an unstable region with nucleate and film boiling occurring alternately, characterized by a decrease in the wall heat flux as the wall temperature increases. This definition of transition boiling is useful when dealing with a fluid that has undergone CHF with bulk subcooling or low vapor qualities. However, when CHF is characterized by film dryout (at high vapor qualities), the flow pattern is liquid droplets dispersed in a bulk vapor. In this case, bulk nucleate boiling is unlikely and the post-dryout heat transfer mechanisms are primarily vapor convection and some form of droplet-wall interaction. Hence, it is appropriate to consider post-dryout heat transfer as a consequence of the fluid condition at CHF rather than in terms of transition and film boiling as defined traditionally.
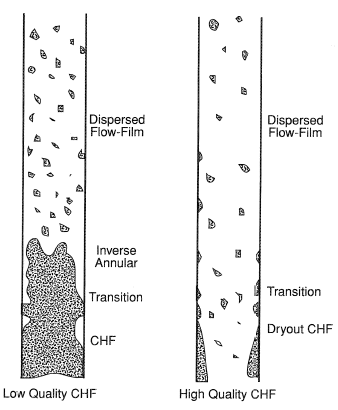
Figure 1 shows typical post-CHF flow conditions for the above two types of postulated CHF and post-dryout situations. If CHF occurs at low vapor fractions, then post-CHF behavior can be expected to be one of inverse annular flow followed by dispersed flow. If the flow rate is also low, then "film boiling" rather than convection will be the dominant heat transfer process. As quality and film thickness increase due to vaporization of the liquid, a decrease in heat transfer capability occurs as the wall temperature increases. This corresponds to the conventional definition of transition boiling.
If CHF occurs at high vapor fractions, the initiating mechanism is dryout of a liquid film at the wall, leading to dispersed drop flow in the post-dryout region. In this situation convection will dominate the heat transfer process, especially if the flow rate is high. Under these conditions, the heat removal capacity of the fluid can either decrease or increase as wall temperature increases, depending on the relative magnitudes of vapor convection and droplet-wall interaction. The usual negative-slope transition boiling portion of the classical boiling curve may not exist in this situation.
Post-dryout heat transfer with bulk subcooling or low vapor qualities occur with inverse annular flow, wherein a central core of liquid is isolated from the heat transfer surface by an annular vapor film. The heat is transferred from the wall to the vapor and subsequently from the vapor to the liquid interphase. If the liquid core is subcooled, a significant fraction of the heat flux is used for sensible heating of the liquid, the balance used to generate vapor at the liquid interphase. If the liquid core is saturated, heat transfer to the interphase is used directly for evaporation. The vapor annulus experiences higher acceleration than the liquid core, leading to substantial slip between the two phases. This velocity differential often causes instability and waves at the liquid interphase, sometimes resulting in entrainment of liquid drops into the vapor flow. These effects enhance heat transfer between the vapor and liquid phases, reducing the tendency for vapor superheating and keeping the vapor temperature close to the local saturation temperature.
Limited experimental measurements of post-dryout heat transfer in the inverse annular film regime have been obtained with refrigerants, cryogenic fluids and water [Bromley et al. (1953), Fung et al. (1979), Groeneveld and Gardiner (1978), Quinn (1965), Ragheb et al. (1981)]. These experimental results indicate the following parametric effects on the post-dryout Heat Transfer Coefficient at the wall (α):
α increases significantly with increases in bulk liquid subcooling, typically 2 to 5% per °C increase in subcooling.
With positive equilibrium vapor Quality (i.e., xe, the equilibrium or thermodynamic quality greater than zero) α decreases with increases in xe at low mass fluxes but increases with xe at high mass fluxes.
The effect of mass flux (
 ) is small or nonexistent for subcooled conditions and low mass fluxes; at higher flow rates and with net vapor quality α increases strongly with increasing mass flux.
) is small or nonexistent for subcooled conditions and low mass fluxes; at higher flow rates and with net vapor quality α increases strongly with increasing mass flux.α increases with increasing system pressure, typically doubling in magnitude for a threefold increase in absolute pressure.
In contrast to behavior in nucleate boiling, α is relatively insensitive to the absolute magnitude of the heat flux.
Some of these parametric effects are illustrated in Figure 2, from Groeneveld (1992).
Methods proposed for prediction of post-dryout heat transfer in this inverse annular flow regime are highly empirical. Groeneveld (1992) reviewed nine different correlations, noting that all were based on limited data and therefore are applicable only in specific ranges of operating parameters. A method that has a wider range of application and converges to predictions for the higher quality disperse regime was suggested by Groeneveld and Rousseau (1983):

where
Tsat = saturation temperature,
Tw = wall temperature,
TG = bulk vapor temperature
and


[Berenson (1961)]

ΔTsub= Tsat − TL ,
ΔTsat = Tw − Tsat,
xa = actual quality.
For inverse annular flow, the vapor phase is often close to thermodynamic equilibrium so that


The above approach considers only convective heat transfer from the wall. Radiation heat transfer is usually small by comparison, but may become significant at high temperatures. In such a case, the additional heat flux due to thermal radiation can be approximated as exchange between parallel infinite surfaces, assuming some effective emissivity for the wall and for the liquid interphase. (See Parallel Plates, Radiative Heat Transfer Between.)
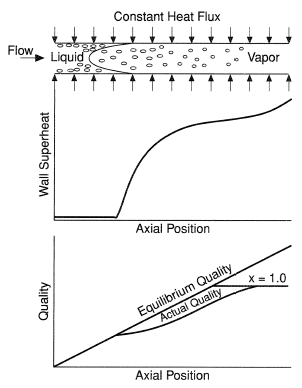
In post-dryout heat transfer, the dispersed flow regime is more often encountered since it can occur over a wide range of vapor qualities, from as low as 0.2 to 1.0. Typical conditions for this regime are illustrated in Figure 3. Before dryout (CHF), fairly low wall superheats are sufficient to sustain convective nucleate boiling. Immediately downstream from the dryout point, the diminished heat transfer coefficient causes the wall temperature to rise to a much higher level, often approaching several hundred degrees Celsius superheat. As noted by several investigators [Chen et al. (1979), Groeneveld and Delorme (1976), Evans et al. (1985)], thermodynamic nonequilibrium often occurs in this regime of two-phase flow, resulting in the actual vapor quality being less than the equilibrium quality (see Figure 3). The liquid phase temperature is normally close to the local saturation temperature. The need to predict the thermohydraulic conditions downstream from the point of dryout may be regarded as an entrance-region problem with the objective of calculating local pressure, equilibrium flow quality, actual flow quality, vapor superheat temperature, heat transfer coefficient at the wall, and either the wall heat flux or wall temperature.
Early experimental investigations [e.g., Bennett et al. (1967)] measured wall temperatures, mass flow, system pressure, inlet equilibrium quality, and wall heat flux, but without measurement of nonequilibrium vapor temperature (TG) or actual quality (xa). In concert with such initial experimental investigations, a simple equilibrium approach was taken to correlate the heat transfer results, based on two major assumptions:
Thermodynamic equilibrium between the two phases at all axial locations.
Heat transfer at the superheated walls primarily by turbulent vapor convection.
The first assumption resulted in taking all fluid temperatures at the local saturation temperature. Consequence of the second assumption was that the wall heat transfer could be represented by a turbulent convective Nusselt Number. This results in a simple model for predicting the post-dryout wall heat flux, given the wall temperature, or vice versa:


where

NuG = vapor convection Nusselt number.
Typical of such equilibrium correlations are those of Dougal-Rohsenow (1963) and Groeneveld (1969). These correlations calculate the wall heat transfer coefficient as a function of mass flux, equilibrium quality, and saturated vapor properties. For example, the Dougall-Rohsenow correlation is

These models are simple and easy to use since they require only information on local conditions at the point of interest, i.e., local wall temperature and equilibrium quality. Compared with the experimental data available in the late 1970s, one finds that these equilibrium correlations predict reasonable trends, though often showing quantitative disagreements of 100 per cent or more.
In recent years, experimental techniques improve to the point that researchers were able to measure superheated vapor temperatures as well as the wall temperature and local heat flux. Typical results as shown in Figure 4, verify that the state of thermodynamic nonequilibrium can exist in this heat transfer regime.
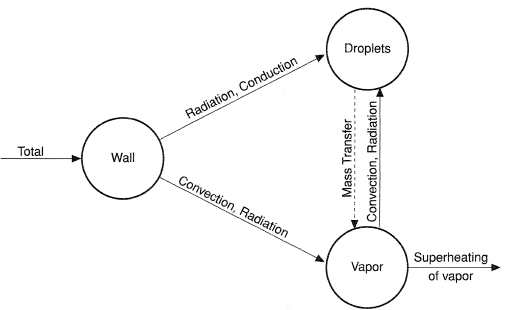
For such conditions, the various interactive mechanisms for heat transfer are illustrated in Figure 5. At high wall superheats, direct liquid contact on the hot surface is minimal and the wall rejects heat mainly by convection and radiation to the vapor and by radiation to the entrained droplets. If thermodynamic nonequilibrium can exist then the vapor could attain a temperature higher than the saturation temperature. In this situation, interfacial heat transfer between the superheated vapor and the droplet surface (which would be at saturation temperature) occur by both convection and radiation. The droplet in turn would be cooled by latent heat of evaporation. By most estimates, the radiative heat transfer is relatively small compared to convective heat transfer. Consequently, the degree of nonequilibrium that occurs is governed by the relative magnitudes of convective heat transfer from the hot wall to the vapor and from the vapor to the entrained droplets. The potential vapor superheat temperature (TG) results from the dynamic balance between these two convective heat transfer processes. Assuming that the equilibrium vapor quality is known at all axial locations from heat balance, the nonequilibrium actual vapor quality (xa) is related to the vapor bulk superheat temperature (TG) by the following relationship

The experimental results of Unal et al. (1988) showed the parametric effects of operating parameters on the vapor superheat (TG − Tsat) and on the temperature difference between wall and vapor (Tw − TG). It is seen from Figure 6 that the vapor superheat decreases with increasing mass flux, increases with increasing wall heat flux, and decreases with actual vapor mass flux. The temperature difference between wall and vapor (Tw − TG) is relatively insensitive to these parameters, though showing a slight trend opposite to that of the vapor superheat.
A number of nonequilibrium models have been proposed in attempts to make better predictions of heat transfer in this dispersed flow regime. One class of models estimates the vapor superheat and wall heat transfer coefficient at any given axial location as functions of only local thermal hydraulic parameters at that location. The CSO model [Chen, Ozkaynak, and Sundaram (1979)] utilizes momentum transfer analogy to calculate a convective heat transfer coefficient between the wall and vapor, together with empirical correlations for the nonequilibrium vapor temperature:


where


and xa from Eq. (7) with

Except for short distances immediately downstream of the dryout point, this wall-vapor convective heat flux represents the major contribution to total wall heat flux. Comparison of this model with experimental measurements show scatter in predicted vapor temperatures but reasonable predictions of wall heat flux.
![Experimental data showing thermodynamic non-equilibrium with superheated vapor [ Chen and Costigan (1992)].](/content/5469/PosHF4.gif)
Figure 4. Experimental data showing thermodynamic non-equilibrium with superheated vapor [ Chen and Costigan (1992)].
![Parametric effects on superheat temperatures [Unal et al. (1988)]. (From Journal of Heat Transfer, 110, Aug. 1988, p. 725, with permission.)](/content/5469/PosHF6.gif)
Figure 6. Parametric effects on superheat temperatures [Unal et al. (1988)]. (From Journal of Heat Transfer, 110, Aug. 1988, p. 725, with permission.)
A more realistic class of models recognizes that the degree of nonequilibrium at any axial location is historically developed by the upstream competitive heat transfer mechanisms (wall-to-vapor versus vapor-to-droplet). One way to treat this situation is to write the conservation equation for the vapor phase, with a volumetric vapor source term (Γ),

where PH = heated perimeter and A = flow area.
If the functional relationship for the source function F and the wall heat transfer coefficient α is known Equations (13) and (14) can be integrated starting at the dryout CHF location to calculate the nonequilibrium actual flow xa at any downstream location z. Various models have been proposed for estimating the vapor source function Γ and for the wall heat transfer coefficient α. An example is the correlation of Saha et al. (1980):







Alternate expressions for this source function Γ have been proposed by Webb and Chen (1982) and Unal et al. (1991).
The above models account only for the direct convective heat transfer between the wall and vapor. While this is usually the dominant mechanism for post-dryout heat transfer in the dispersed flow regime, there are experimental indications that direct heat transfer from the wall to impinging liquid drops may be significant at short distances downstream of the dryout location [Unal et al. (1988), Cokmez-Tuzla et al. (1993)]. Since models for this wall-liquid transfer mechanism are in early stages of development, current capability can only account for wall-vapor convection as described above. Radiative heat transfer needs to be considered also, but is found to be less than 10 per cent of total heat transfer in most applications.
REFERENCES
Berenson, P. J. (1961) Film boiling heat transfer from a horizontal surface, J. of Heat Transfer, 83, 351-358.
Bromley, L. A., LeRoy, N. R., and Robbers, J. A. (1953) Heat transfer in forced convective film boiling, Ind. Eng., Chem., 45, 2639-2646.
Chen, J. C, Ozkaynak, F. T., and Sundaram, R. K. (1979) Vapor heat transfer in post~CHF region including the effect of thermodynamic nonequilibrium. Nuclear Eng. and Design, 51, 143. DOI: 10.1016/0029-5493(79)90086-4
Chen, J. C, Sundaram, R. K., and Ozkaynak, F. T. (1977) A Phenomenological Correlation for Post-CHF Heat Transfer, NUREG-0237, U.S. Nuclear Regulatory Commission. National Technical Information Service, Springfield, VA, USA.
Chen, J. C. and Costigan, G. (1992) Review of Post-dryout heat transfer in dispersed two-phase flow, Post Dryout Heat Transfer, Multiphase Science and Technology, G. F. Hewitt, J. M. Delhaye, N. Zuber, Eds. CRC Press, London, 1-37.
Dougall, R. S. and Rohsenow, W. M. (1963) Film Boiling on the Inside of Vertical Tubes with Upward Flow of the Fluid at Low Qualities, MIT Report No. 9079-26.
Evans, D., Webb, S. W., and Chen, J. C. (1985) Axially varying vapor superheats in convective film boiling, J. of Heat Transfer, 107, 3, 663-669
Fung, K. K., Gardiner, S. R. M. and Groeneveld, D. C. (1979) Subcooled and low quality flow film boiling of water at atmospheric pressure, Nucl. Eng., and Design, 55, 51-57. DOI: 10.1016/0029-5493(79)90143-2
Groeneveld, D. C. and Delorme, D. J. (1976) Prediction of thermal nonequilibrium in the post-dryout regime, Nuclear Eng. and Design, 36, 17. DOI: 10.1016/0029-5493(76)90138-2
Groeneveld, D. C. and Rousseau, J. C. (1983) CHF and Post-CHF heat transfer: An assessment of prediction methods and recommendations for reactor safety codes, Proceedings NATO Meeting on Advances in Two-Phase Flow and Heat Transfer, 1, 203-239, NATO ASI Series, Series E. No. 63, Nijhoff Publishers.
Groeneveld, D. C. (1992) A review of inverted annular and low quality film boiling, Post-Dryout Heat Transfer, G. F. Hewitt, J. M. Delhaye and N. Zuber Eds., CRC Press, London, 327-366.
Groeneveld, D. C. (1969) An Investigation of Heat Transfer in the Liquid Deficient Regime, Report AECL-3281.
Groeneveld, D. C. and Gardiner, S. R. M. (1978) A method of obtaining flow film boiling data for subcooled water, Int. J. Heat and Mass Transfer, 21, 664-665. DOI: 10.1016/0017-9310(78)90068-6
Quinn, E. P. (1965) Forced Flow Transition Boiling Heat Transfer from Smooth and Finned Surfaces, GEAP-4786.
Ragheb, H. S., Cheng, S. C, and Groeneveld, D. C. (1981) Observations in transition boiling of subcooled water under forced convective conditions, Int. J. of Heat and Mass Transfer, 24, 7, 1127-1137. DOI: 10.1016/0017-9310(81)90162-9
Unal, C., Tuzla, K., Badr, O., Neti, S., and Chen, J. C. (1988) Parametric trends for post-CHF heat transfer in rod bundles, J. of Heat Transfer, 110, 721-727.
Unal, C., Tuzla, K., Cokmez-Tuzla, A. and Chen, J. C. (1991) Vapor generation rate model for dispersed drop flow, Nuclear Eng. and Design, 125, 161-173. DOI: 10.1016/0029-5493(91)90075-S
Webb, S. W. and Chen, J. C. (1982) Vapor generation rate in non-equilibrium convective film boiling, Proc. 7th International Heat Transfer Conf, 4, 437-442, Munich.
References
- Berenson, P. J. (1961) Film boiling heat transfer from a horizontal surface, J. of Heat Transfer, 83, 351-358.
- Bromley, L. A., LeRoy, N. R., and Robbers, J. A. (1953) Heat transfer in forced convective film boiling, Ind. Eng., Chem., 45, 2639-2646. DOI: 10.1021/ie50528a027
- Chen, J. C, Ozkaynak, F. T., and Sundaram, R. K. (1979) Vapor heat transfer in post~CHF region including the effect of thermodynamic nonequilibrium. Nuclear Eng. and Design, 51, 143. DOI: 10.1016/0029-5493(79)90086-4
- Chen, J. C, Sundaram, R. K., and Ozkaynak, F. T. (1977) A Phenomenological Correlation for Post-CHF Heat Transfer, NUREG-0237, U.S. Nuclear Regulatory Commission. National Technical Information Service, Springfield, VA, USA.
- Chen, J. C. and Costigan, G. (1992) Review of Post-dryout heat transfer in dispersed two-phase flow, Post Dryout Heat Transfer, Multiphase Science and Technology, G. F. Hewitt, J. M. Delhaye, N. Zuber, Eds. CRC Press, London, 1-37.
- Dougall, R. S. and Rohsenow, W. M. (1963) Film Boiling on the Inside of Vertical Tubes with Upward Flow of the Fluid at Low Qualities, MIT Report No. 9079-26.
- Evans, D., Webb, S. W., and Chen, J. C. (1985) Axially varying vapor superheats in convective film boiling, J. of Heat Transfer, 107, 3, 663-669
- Fung, K. K., Gardiner, S. R. M. and Groeneveld, D. C. (1979) Subcooled and low quality flow film boiling of water at atmospheric pressure, Nucl. Eng., and Design, 55, 51-57. DOI: 10.1016/0029-5493(79)90143-2
- Groeneveld, D. C. and Delorme, D. J. (1976) Prediction of thermal nonequilibrium in the post-dryout regime, Nuclear Eng. and Design, 36, 17. DOI: 10.1016/0029-5493(76)90138-2
- Groeneveld, D. C. and Rousseau, J. C. (1983) CHF and Post-CHF heat transfer: An assessment of prediction methods and recommendations for reactor safety codes, Proceedings NATO Meeting on Advances in Two-Phase Flow and Heat Transfer, 1, 203-239, NATO ASI Series, Series E. No. 63, Nijhoff Publishers.
- Groeneveld, D. C. (1992) A review of inverted annular and low quality film boiling, Post-Dryout Heat Transfer, G. F. Hewitt, J. M. Delhaye and N. Zuber Eds., CRC Press, London, 327-366.
- Groeneveld, D. C. (1969) An Investigation of Heat Transfer in the Liquid Deficient Regime, Report AECL-3281.
- Groeneveld, D. C. and Gardiner, S. R. M. (1978) A method of obtaining flow film boiling data for subcooled water, Int. J. Heat and Mass Transfer, 21, 664-665. DOI: 10.1016/0017-9310(78)90068-6
- Quinn, E. P. (1965) Forced Flow Transition Boiling Heat Transfer from Smooth and Finned Surfaces, GEAP-4786.
- Ragheb, H. S., Cheng, S. C, and Groeneveld, D. C. (1981) Observations in transition boiling of subcooled water under forced convective conditions, Int. J. of Heat and Mass Transfer, 24, 7, 1127-1137. DOI: 10.1016/0017-9310(81)90162-9
- Unal, C., Tuzla, K., Badr, O., Neti, S., and Chen, J. C. (1988) Parametric trends for post-CHF heat transfer in rod bundles, J. of Heat Transfer, 110, 721-727.
- Unal, C., Tuzla, K., Cokmez-Tuzla, A. and Chen, J. C. (1991) Vapor generation rate model for dispersed drop flow, Nuclear Eng. and Design, 125, 161-173. DOI: 10.1016/0029-5493(91)90075-S
- Webb, S. W. and Chen, J. C. (1982) Vapor generation rate in non-equilibrium convective film boiling, Proc. 7th International Heat Transfer Conf, 4, 437-442, Munich.
![Parametric effects for inverse annular heat transfer [Groeneveld (1992)].](/content/5469/PosHF2.gif)